Summary
-
1.
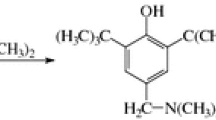
Homogeneous destructive hydrogenation of three isomeric butylphenols at 440° and high hydrogen pressures was investigated. The reaction rate decreased in the series o-n-butylphenol> p-n-butylphenol>p-sec-butylphenol. The difference in the rate of total conversion was determined predominantly by the stage in which there was thermal cleavage of the weakest bond of the alkyl group.
-
2.
A rise in hydrogen pressure accelerated the process of homogeneous destructive hydrogenation of butylphenols.
-
3.
The weakest bonds in n-butylphenols were apparently the β and γ C-C bonds in the alkyl chain; the same was true also for para sec-butylphenol.
-
4.
Comparison of the results obtained led to the conclusion that depending on the increase in the number of carbon atoms in the side chain of the alkylphenol, thermal decomposition processes start to prevail to a greater extent over reactions leading to cleavage of the entire alkyl radical from the aromatic nucleus.
Similar content being viewed by others
Literature cited
A. E. Gavrilova, M. G. Gonikberg, V. T. Aleksanyan, and Kh. E. Sterin, Izv. AN SSSR, Otd. khim. n. 472 (1958).
M. G. Gonikberg and Li Huan-nyan'. Izv. AN SSSR, Otd. khim. n. 498 (1960), 491 (1961).
V. Rice and C. Harden, J. Amer. Pharm. Assoc.25, 7 (1936).
W. Steinkopf and Th. Höpner., J. prakt. chem.113, 140 (1926).
O. Kruber and A. Schmift, Ber.64, 2272 (1931).
M. G. Gonikberg and V. E. Nikitenkov, Dokl. AN SSSR,102, 949 (1955).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gavrilova, A.E., Gonikberg, M.G. Investigation of the homogeneous destructive hydrogenation of butylphenols under hydrogen pressure. Russ Chem Bull 10, 1576–1579 (1961). https://doi.org/10.1007/BF00906158
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00906158