Abstract
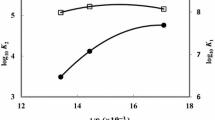
Complex formation between uranyl ion (UO 2+2 ) andl-proline has been studied at 30°C and ionic strength 0.1M (KCl) using polarographic technique. At pH 3.0, analysis of the results by simple graphical andDe Ford andHume methods revealed the formation of two complex species, [UO2(Pro)]+ and [UO2(Pro)2]. The formation of the second species become evident only at higher proline concentrations (>0.25M).
Similar content being viewed by others
Literatur
J. P. Greenstein undM. Winitz, Chemistry of amino acids, Vol. 1, S. 569. New York: Wiley, 1966.
O. Farooq, A. U. Malik, N. Ahmad undS. M. F. Rahman, J. Electroanal. Chem.24, 464 (1970);K. K. Girdhar, K. K. Vaid undP. S. Relan, J. Indian Chem. Soc.47, 715 (1970);G. M. Sergeev undI. A. Korshunov, Radiokhimiya15, 621 (1973).
A. M. Bond, Coord. Chem. Rev.6, 377 (1971).
J. Kuta undE. Yeager, J. Electroanal. Chem.31, 119 (1971) und dort zit. Literatur.
W. E. Harris undI. M. Kolthoff, J. Amer. Chem. Soc.69, 446 (1947).
Kabir-ud-Din, J. Kuta undL. Pospisil, Electrochim. Acta22, 1109 (1977).
W. E. Harris undI. M. Kolthoff, J. Amer. Chem. Soc.67, 1484 (1945).
D. D. De Ford undD. N. Hume, J. Amer. Chem. Soc.73, 5321 (1951).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kabir-ud-Din, Khan, I.A. Polarographische Untersuchungen an Uranyl-L-Prolin-Komplexen. Monatshefte für Chemie 109, 1343–1348 (1978). https://doi.org/10.1007/BF00906047
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00906047