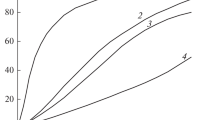
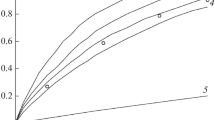
Conclusions
-
1.
The decomposition of 3,3-dinitrobutanol-2 in water was studied at 25° and ionic strength of the solution 0.1.
-
2.
The equilibrium constant of the decomposition of 3,3-dintrobutanol-2 into acetaldehyde and dinitro-ethane was determined.
-
3.
The second-order rate constant of the interaction of acetaldehyde and the dinitroethane anion was determined.
Similar content being viewed by others
Literature cited
V. M. Belikov and Yu. I. Belokon', Collection: Reactivity of Organic Compounds [in Russian], Tartu,2, No. 2 (1965), p. 211.
T. N. Hall, J. Organ. Chem.,30, 3157 (1965).
R. Kaplan and H. Schechter, J. Amer. Chem. Soc.,83, 3535 (1961).
T. N. Hall, J. Organ. Chem.,29, 3587 (1964).
L. A. Yanovskaya and V. M. Belikov, Izv. AN SSSR, Ser. Khim.,1965, 1363.
E. A. Gugenheim, Phil. Mag.,7, No. 2, 538 (1926).
R. P. Bell, The Proton in Chemistry, Ithaca (1959), p. 134.
A. Albert and E. Sergeant, Ionization Constants of Acids and Bases [Russian translation], “Khimiya,” Moscow (1964), p. 20.
F. P. Bell and J. C. Clunie, Trans. Faraday Soc.,48, 439 (1952).
Author information
Authors and Affiliations
Additional information
The article is published on the basis of a resolution of the Conference of Editors-in-Chief of the Journals of the Academy of Sciences of the USSR, of July 12, 1962, as the dissertation work of Yu. N. Belokon'.
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 3, pp. 528–532, March, 1967.
Rights and permissions
About this article
Cite this article
Belokon', Y.N., Belikov, V.M. Kinetics of the decomposition of 3,3-dinitrobutanol-2 in water under the action of bases. Russ Chem Bull 16, 512–515 (1967). https://doi.org/10.1007/BF00905982
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00905982