Conclusions
-
1.
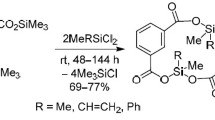
A study was made of the cohydrolysis reaction of phenyldichlorosilane with triethylchlorosilane and dialkyl(aryl)dichlorosilanes. The reaction proceeds with the formation of cohydrolysis products and the products of separate hydrolysis.
-
2.
The cohydrolysis of dialkyl(aryl)dichlorosilanes with phenyldichlorosilane gives up to 73% of distillable cyclic compounds and undistillable oligomers.
-
3.
The cohydrolysis of phenyldichlorosilane with triethylchlorosilane led to the isolation of some new phenylhydrosiloxanes with triethylsilyl end groups of general formula (C2H5)3Si[O-Si(C6H5)H]n-OSi(C2H5 3, where n=1–4, while the cohydrolysis of phenyldichlorosilane with dialkyl(aryI)dichlorosilanes led to the isolation of seven new cyclic alkylarylcyclosiloxanes.
Similar content being viewed by others
Literature cited
W. F. Mivelas, Dissertation, Indiana University (1956).
M. C. Harvey, W. H. Nebergall, and J. S. Peake, J. Am Chem. Soc.79, 1437 (1957).
K. A. Andrianov, A. I. Nogaideli, L. M. Khananashvili, and L. I. Nakaidze, Soobshch. Akad. Nauk Gruz. SSR,48, 323 (1967).
V. Bazant, V. Chvalovsky, and J. Rathousky, Organosilicon Compounds, Prague (1965).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 4, pp. 828–832, April, 1968.
Rights and permissions
About this article
Cite this article
Andrianov, K.A., Nogaideli, A.I., Khananashvili, L.M. et al. Cohydrolysis reaction of phenyldichlorosilane with triethylchlorosilane and dialkyl(aryl)dichlorosilanes. Russ Chem Bull 17, 793–796 (1968). https://doi.org/10.1007/BF00905755
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00905755