Abstract
Dissolution of zinc and nickel ferrites were previously found2,3 to conform to the equations:
x-solubility (%),t=time (min),s 0=initial specific surface (m 2·g−1),k-rate constant independent of specific surface,K=apparent rate constant dependent on specific surface (min−1).
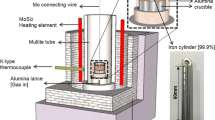
The aim of this work was to check the applicability of these equations to the dissolution of nickel—zinc ferrites. The experimental results obtained for 3 mixed ferrites (Ni0.3Zn0.7Fe2O4, Ni0.5Zn0.5Fe2O4, Ni0.7Zn0.3Fe2O4) revealed that kinetics of their dissolution in HCl, HNO3 and their mixtures also conform to the equations stated above.
Similar content being viewed by others
Literatur
Wł. Wolski undI. Kozłowska, J. Japan Soc. Powder and Powder Metal.18, 8 (1970).
I. Okońska-Kozłowska, Mh. Chem.103, 1585 (1972).
F. J. Verwey, P. W. Haaymann undF. C. Romeijn, J. Chem. Phys.15, 182 (1947).
Author information
Authors and Affiliations
Additional information
Mit 3 Abbildungen
I. Okońska-Kozlowska, Teil der Dissertation, Poznań 1969.
Rights and permissions
About this article
Cite this article
Okońska-Kozlowska, I. Löslichkeit von Nickel-Zink-Ferriten in Säuren. Monatshefte für Chemie 104, 1040–1044 (1973). https://doi.org/10.1007/BF00903920
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00903920