Abstract
Study of the ageing process of amorphous ferric hydroxide has shown that the presence of traces of Al3+ can drastically change the course of the reaction under certain conditions.
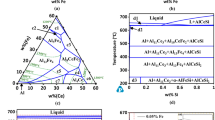
Al3+ traces delay crystallization, an effect which is dependant on the pH of the solution in which the originally amorphous gel is boiled. With increasing pH they favour the formation of the α-F2O3-phase, whereby the α-FeOOH modification, the formation of which should otherwise be favoured by the dehydrative conditions employed, is not formed at all. Inhibition of the formation of the α-FeOOH phase is already crystallographically detectable at a molar ratio of Al2O3/Fe2O3=0.001. The complete disappearance of this phase at pH=10 of the ageing medium is found at the molar ratio Al2O3/Fe2O3=0.03. At higher Al3+ concentrations the amorphous gel is converted into α-Fe2O3. In the range 10–20 mol% Al2O3, crystallization is strongly inhibited. Further increase in the Al3+ content favours the formation of Bayerite-phase, which then at 50 mol% Al predominantes, alongside poorly crystalline α-Fe2O3.
For Al2O3/Fe2O3=0.55, three phases are present, hydrohematite, bayerite and boehmite. With still greater amounts of Al3+ only the boehmite modification is found.
By stepwise crystallographic study of the progressive dehydration the duration of reaction has been established for ferric hydroxide both in the pure state and when doped with Al3+.
Similar content being viewed by others
Literatur
A. Neuhaus undH. Heide, Ber. dtsch. Keram. Ges.42, 167 (1965).
K. Wefers, Erz, Metall20, 71 (1967).
J. White, Proc. 2nd Internat. Symp. ICSOBA, Vol. 2, 195 (1971).
R. Fricke undHüttig, Oxyde und Oxydhydrate, Leipzig 1937.
A. Kuhn, Kolloidchemisches Taschenbuch, 5. Aufl., S. 227–315. Leipzig 1960.
F. Bernal, D. Dasgupta undA. Mackay, Clay Min. Bull.4, 15 (1959).
K. Torkar, Mh. Chem.94, 110 (1963), sowie alle in den Monatsheften für Chemie in den Jahren 1960–1963 veröffentlichten Mitteilungen.
A. Krause, Z. anorg. allgem. Chem.219, 213 (1937).
A. Lewandowski, Roczn. Chem.23, 256 (1949).
R. Thiel, Z. anorg. allgem. Chem.326, 70 (1963).
G. Montel, C. r. hebdomad. Sé. Acad. Sci.250, 716 (1960).
S. Caillére, L. Gatineau undS. Henin, C. r. hebdomad. Sé. Acad. Sci.250, 3677 (1960).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wolska, E. Zur Kenntnis der künstlichen Alterung aluminiumhaltiger Eisen(III)-hydroxide. Monatshefte für Chemie 106, 905–913 (1975). https://doi.org/10.1007/BF00900870
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00900870