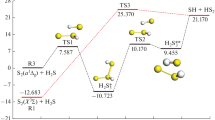
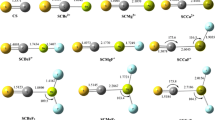
Abstract
It is well known that as the valency of the sulfur atom in sulfur-containing compounds increases, so does the population of vacant 3d orbitals of sulfur. The possibilities of an experimental approach to the problem of the electron population of vacant 3d orbitals are extremely limited since each experimental fact is, as a rule, due to a combination of a whole number of interactions and it is practically impossible to distinguish in it that part which is associated precisely with a vacant antibonding orbital (AO). In a number of papers the contributions from the latter interactions are neglected and the effect is attributed entirely to vacant orbitals. Since theoretical investigations on particular properties in a number of molecules within the same approximation make it possible to convey the variations of these properties qualitatively, we have endeavored, with the example of calculations on the electronic structure and spectra, in sulfur-containing compounds with different sulfur atom valencies, to follow the changes in these properties while at the same time revealing the effect of vacant 3d orbitals of the sulfur atom.
Similar content being viewed by others
Literature cited
P. Mollere and H. Bock, J. Organometallic Chem.,61, 127 (1973).
V. I. Vovna and F. I. Vilesov, in: Progress in Photonics [in Russian], Leningrad State Univ. (1975).
R. McDiarmid, J. Chem. Phys.,61, 274 (1974).
A. Mangini, M. Pallotti, M. Tiecco, et al., Int. J. Sulfur Chem.,A2, 69 (1972).
O. V. Sverdlova, Electron Spectra in Organic Chemistry [in Russian], Khimiya, Leningrad (1973), p. 125.
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Vysshikh Uchebnykh Zavedenii, Fizika, No. 5, pp. 19–22, May, 1978.
Rights and permissions
About this article
Cite this article
Khaustova, V.I. Theoretical investigation of electronic structure and spectra of dimethyl sulfide, sulfoxide, and sulfone. Soviet Physics Journal 21, 564–567 (1978). https://doi.org/10.1007/BF00890964
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00890964