Summary
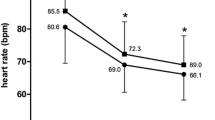
The results of an open prospective study that evaluated the long-term clinical safety of nicorandil are presented. This study included 199 patients with severe chronic stable angina treated over a 1-year period. The most often reported adverse event was headache, which was responsible for most of the study withdrawals due to clinical intolerance (9.6%). When using a progressive titration scheme, this incidence was substantially reduced to 2.7%. As with other less frequent adverse events (dizziness, gastrointestinal disorders), headaches were reported as being mild to moderate in severity, were experienced during the first days of treatment, and, if treatment was maintained, usually resolved within a few days. The incidence of adverse events was not modified when nicorandil was given in combination with a beta-blocker, a calcium antagonist, or both agents. Cardiovascular safety was satisfactory and laboratory parameters were not altered. At the end of the study, 70% of patients were maintained on nicorandil. These results are in agreement with those reported from the nicorandil safety database, which gathered 1152 patients treated by nicorandil, including those of the present study. In comparative studies of nicorandil versus beta-blockers, calcium antagonists, or nitrates, the overall incidence of adverse events was no different between the two treatment groups, although the safety profile differed according to the drug category. During the course of several clinical studies, nicorandil was associated with antihypertensive, antidiabetic, hypolipemic, or other antianginal agents; these combinations did not increase the incidence of adverse events or study withdrawals. In all long-term clinical studies using doses up to 80 mg/day, nicorandil has had a favorable cardiovascular safety profile. There were no meaningful changes in heart rate and blood pressure, and ECG at rest and during exercise did not show rhythm or conduction disorders or QT- and ST-segment modification. Up to now, there has been no evidence that nicorandil exerts a negative inotropic effect. The nicorandil safety profile was not different in patients aged over 65 years (33%). Finally, nicorandil did not affect the usual laboratory parameters, including blood potassium and glucose levels, and the blood lipid profile. Therefore, nicorandil, alone or in combination, appears to be a safe, active antianginal agent for the long-term treatment of a wide range of patients with chronic stable angina.
Similar content being viewed by others
References
Waters D. Proischemic complications of dihydropyridine calcium channel blockers.Circulation 1991;84:2598–2600.
Duty S, Weston AH. Potassium channel openers. Pharmacological effects and future uses.Drugs 1990;40:785–791.
Krumenacker M, Roland E. Clinical profile of nicorandil: An overview of its hemodynamic properties and therapeutic efficacy.J Cardiovasc Pharmacol 1992;20(Suppl 3):S93-S102.
Roland E. Safety profile of an anti-anginal agent with potassium channel opening activity.Eur Heart J 1993;14(Suppl B):48–52.
Krumenacker M. Clinical documentation: Overall summary. Nicorandil registration file. Rhône-Poulenc Rorer report no. 105243, 1990, data on file.
Guermonprez JL, Blin P, Peterlongo F. A double-blind comparison of the long-term efficacy of a potassium channel opener and a calcium antagonist in stable angina pectoris.Eur Heart J 1993;14(Suppl B):30–34.
Ulvenstam G, Diderholm E, Frithz G, et al. Antianginal and antüschemic efficacy of nicorandil compared with nifedipine in patients with angina pectoris and coronary heart disease: A double blind, randomized, multicenter study.J Cardiovasc Pharmacol 1992;20(Suppl 3):S67-S73.
Doring G. Antianginal and anti-ischemic efficacy of nicorandil in comparison with isosorbide-5-mononitrate and isosorbide dinitrate: Results from two multicenter, double-blind, randomized studies with stable coronary heart disease patients.J Cardiovasc Pharmacol 1992;20(Suppl 3):S74-S81.
Frydman A. Pharmacokinetic profile of nicorandil in humans: An overview.J Cardiovasc Pharmacol 1992;20(Suppl 3):S34–44.
Escande D, Cavero I. Potassium channel openers in the heart. In: Escande D, Standen N, eds.K + Channels in Cardiovascular Medicine. France: Springer-Verlag 1993:225–244.
Wilde AAM, Janse MJ. Electrophysiological effects of ATP sensitive potassium channel modulation: Implications for arrhythmogenesis.Cardiovasc Res 1994;28:16–24.
Horfmann D, Opie LH. Potassium channel blockade and acute myocardial infarction: Implications for the management of the non-insulin requiring diabetic patient.Eur Heart J 1993;14:1585–1589.
Yanagisawa T, Taira N. Effect of 2-nicotinamidethyl nitrate (SG-75) on membrane potentials of canine Purkinje fibers.Jpn J Pharmacol 1981;31:409–417.
Habuchi Y, Nishimura M, Watanabe Y. Electrophysiologic effects of nicorandil, a new antianginal agent, on action potentials and membrane currents of rabbit atrioventricular node.Naunyn Schmiedebergs Arch Pharmacol 1987;335:567–574.
Taira N. Nicorandil as a hybrid between nitrates and potassium channel activators.Am J Cardiol 1989;63:18J-24J.
Kojima S, Ishikawa S, Ohsawa K, et al. Determination of effective and safe dose for intracoronary administration of nicorandil in dogs.Cardiovascular Res 1990;24:727–732.
Kempsford RD, Hawgood BJ. Assessment of the antiarrhythmic activity of nicorandil during myocardial ischemia and reperfusion.Eur J Pharmacol 1989;163:61–68.
Gross GJ, Warltier DC, Hardman HF. Comparative effects of nicorandil, a nicotinamide nitrate derivative, and nifedipine on myocardial reperfusion injury in dogs.J Cardiovasc Pharmacol 1987;10:535–542.
Endo T, Nejima J, Kiuchi K, et al. Reduction of size of myocardial infarction with nicorandil, a new antianginal drug, after coronary artery occlusion in dogs.J Cardiovasc Pharmacol 1988;12:587–592.
Lamping KA, Christensen CW, Pelc LR, et al. Effects of nicorandil and nifedipine on protection of ischemic myocardium.J Cardiovasc Pharmacol 1984;6:536–542.
Cohen-Solal A, Jaeger P, Bouthier J, et al. Hemodynamic action of nicorandil in congestive heart failure.Am J Cardiol 1989;63:44J-48J.
Tice FD, Binkley PF, Cody RJ, et al. Hemodynamic effect of oral nicorandil in congestive heart failure.Am J Cardiol 1990;65:1361–1367.
Yokota M, Horisawa T, Iwase M, et al. Effects of a new vasodilator, nicorandil, on exercise-induced impairment of left ventricular function in patients with old myocardial infarction.J Cadiovasc Pharmacol 1987;10(Suppl 8):S116-S122.
Silke B, Verma SP, Ali MS, et al. Effects of nicorandil on left ventricular hemodynamics and volume at rest and during exercise-induced angina pectoris.Am J Cardiol 1989;63:49J-55J.
Thormann J, Schlepper M, Kramer W, et al. Effectiveness of nicorandil (SG-75), a new long-acting drug with nitroglycerin effects, in patients with coronary artery disease: Improved left ventricular function and regional wall motion and abolition of pacing-induced angina.J Cardiovasc Pharmacol 1983;5:371–377.
Kambara H, Nakamura Y, Takami S, et al. Beneficial effects of nicorandil on cardiovascular hemodynamics and left ventricular function.J Cardiovasc Pharmacol 1987;10(Suppl 8):S104-S108.
Kambara H, Takami S, Nakamura Y, et al. Effects of intravenous administration of nicorandil on cardiovascular hemodynamics and left ventricular function.Am J Cardiol 1989;63:56J-60J.
Chikamatsu H, et al. Electrophysiologic effects of nicorandil, an anti-anginal drug in man.Acta Pharmacol Toxicol 1986;59(Suppl V).
Mitrovic V, Neuss H, Kindler M. Electrophysiological effect of a vasodilatation induced by nicorandil.Herz Kreisl 1986;18:403–408.
Tamby JF. Nicorandil registration file. Rhône-Poulenc Rorer 1992. Data on file.
Saito S, Tamura Y, Moriuchi M, et al. Comparative efficacy and safety of nitroglycerin, verapamil and nicorandil during coronary angioplasty.J Am Coll Cardiol 1991;17(Suppl A):377.
Williams AJ, Lee TH, Cochrane GM, et al. Attenuation of nocturnal asthma by cromakalim.Lancet 1990;336:334–336.
Garrino MG, Plant TD, Henquin JC. Effects of putative activators of K+ channels in mouse pancreatic β-cells.Br J Pharmacol 1989;98:957–965.
Dune MJ. Effects of pinacidil, RP 49356 and nicorandil on ATP-sensitive channels in insulin-secreting cells.Br J Pharmacol 1990;99:487–492.
Pratz J, Mondot S, Montier F, et al. Effects of the K+ channel activators, RP 52891, cromakalim and diazoxide on the plasma insulin level, plasma renin activity and blood pressure in rats.J Pharmacol Exp Ther 1991;258:216–222.
Author information
Authors and Affiliations
Additional information
Coordinator of the investigators' group for study SG8035; Drs. Akoun, Neuilly S/Seine; Backouche, Paris; Bar-David, Paris; Belliard, Paris; Berdellou, Aulnay Sous-Bois; Botherel, Arcueil; Bouchain, Guerville; Buzzi, Paris; Camus, Vincennes; Chaouat, Paris; Chokron, Paris; Darsin, Aulnay Sous-Bois; Degroote, Bagneux; Demelle, Paris; Dubray, Pontoise; Dumoulin, Courbevoie; Duval, Pontoise; Fitoussi, Paris; Giraudel, Marly La Forêt; Gryner, Paris; Haddad, Pontoise; Hildesheim, Paris; Huet-Maillet, Paris; Jeremiasz, Paris; Koubi, Pré Saint-Gervais; Leblond, Arcueil; Rondou, La Courneuve; Smadja, Paris; Thébaut, Sarcelles; Trinh-Trinh, Vigneux S/Seine; Yvenou, Paris).
Rights and permissions
About this article
Cite this article
Witchitz, S., Darmon, JY. Nicorandil safety in the long-term treatment of coronary heart disease. Cardiovasc Drug Ther 9 (Suppl 2), 237–243 (1995). https://doi.org/10.1007/BF00878471
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00878471