Summary
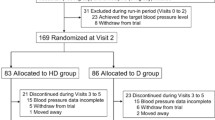
The aim of this double-blind parallel-group study was to compare the effects of doxazosin, a selective alpha1-adrenoceptor antagonist with a long plasma half-life, with nitrendipine, a long-acting calcium-entry blocking drug. Following a 4-week placebo period, 26 patients with mild-to-moderate essential hypertension were randomly allocated to treatment with either doxazosin (n=12) or nitrendipine (n=14). Over a period of 10 weeks, doses were titrated to obtain a standing diastolic pressure below 90 mmHg. Thereafter, optimal doses were continued for another 4 weeks. Both drugs were administered once daily; median doses were 4 mg/day for doxazosin and 10 mg/day for nitrendipine. During the titration period three patients in the doxazosin group and one in the nitrendipine group dropped out from the study; one patient on doxazosin was considered a nonresponder. Twenty-one patients completed the study. The percentage of patients showing an adequate hypotensive effect (standing diastolic pressure below 90 mmHg) at the end of the study was similar in the two groups (42% vs. 50% in the intention-to-treat analysis and 56% vs. 54% in the per-protocol analysis). Casual, basal, and standing blood pressure and heart rate did not differ between groups throughout the study; serum lipids and blood glucose remained unchanged. We conclude that doxazosin and nitrendipine given as monotherapy are equally effective in mild to moderate hypertension.
Similar content being viewed by others
References
Van Zwieten PA, Van Meel JCA, Timmermans PBMWM. Pharmacology of calcium entry blockers: Interaction with vascular alpha-adrenoceptors.Hypertension 1983;5(Suppl II):8–17.
Young RA, Brogden RN. Doxazosin. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic efficacy in mild or moderate hypertension.Drugs 1988;35:525–541.
De Leeuw PW, Ligthart JJ, Smout AJPM, Birkenhäger WH. Within patient comparison of prazosin and UK-33,274. A new alpha-adrenoceptor-antagonist.Eur J Clin Phamacol 1982;23:397–401.
Lund-Johansen P, Omvik P, Haugland H. Acute and chronic haemodynamic effects of doxazosin in hypertension at rest and during exercise.Br J Clin Pharmacol 1986;21(Suppl 1):45–54.
Cubeddu LX, Fuenmayor N, Caplan N, Ferry D. Clinical pharmacology of doxazosin in patients with essential hypertension.Clin Pharmacol Ther 1987;41:439–449.
Goa KL, Sorkin EM. Nitrendipine. A review of its pharmacodynamic and pharmacokinetic properties and therapeutic efficacy in the treatment of hypertension.Drugs 1987;33:123–155.
Vincent J, Elliott HL, Meredith PA, Reid JL. Doxazosin, an a1-adrenoceptor antagonist: Pharmacokinetics and concentration-effect relationships in man.Br J Clin Pharmacol 1983;15:719–725.
Hansson L, Andrén L, Orö L, Ryman T. Pharmacokinetic and pharmacodynamic parameters in patients treated with nitrendipine.Hypertension 1983;5(Suppl II):25–28.
Cubeddu LX, Pool JL, Bloumfield R, et al. Effect of doxazosin monotherapy on blood pressure and plasma lipids in patients with essential hypertension.Am J Hypertens 1988;1:158–167.
Taylor SH, Le PS, Sharna SK. A comparison of doxazosin and enalapril in the treatment of mild and moderate essential hypertension.Am Heart J 1988;116:1820–1825.
Fodor JG, Chockalingam A. Treatment of mild hypertension.J Cardiovasc Pharmacol 1991;18(Suppl 1):S25-S29.
Lederle RM, Klaus D, Wilk S, Knaup G. Low-dose nitrendipine in mild hypertension: A double-blind, placebo-controlled, comparative study.J Cardiovasc Pharmacol 1991;18(Suppl 1):S48-S50.
Nechwatal W, Berger J, Blumrich W, et al. A double-blind comparative study of doxazosin and nitrendipine in patients with mild to moderate essential hypertension.Am Heart J 1988;116:1806–1814.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cosenzi, A., Waltman, F.L., van Es, P.N. et al. Doxazosin versus nitrendipine: A double-blind comparative study in patients adhering to a sodium-restricted diet. Cardiovasc Drug Ther 8, 473–477 (1994). https://doi.org/10.1007/BF00877925
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00877925