Abstract
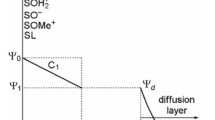
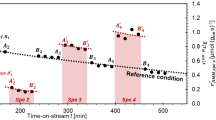
Paul Schindler's early work on the acid-base chemistry of oxides was instrumental for the development of the concept of surface complexation. This approach has not only been important in establishing a theory on the adsorption of metal ions and ligands as a function of pH and solution variables, but has become essential in establishing surface speciation (coordinative structural and electronic arrangement at the solidwater interface) which in turn determines surface reactivity. The factors that affect dissolution of Fe(III) (hydr)oxides and inhibition of dissolution are discussed. A few examples for the inhibition of reductive and ligand-promoted dissolution by binuclear complexes of oxoanions (phosphate, borate) and of protonpromoted dissolution by Cr(III) are given.
Similar content being viewed by others
References
Biber, M. Dos Santos Afonso, M., Stumm W. (submitted) The coordination chemistry of weathering: IV. Inhibition of the dissolution of oxide minerals. Geochim. Cosmochim. Acta.
Blesa, M.A., Regazzoni, A.E., Stumm, W., (in preparation).
Bondietti, G., Sinniger, J., Stumm, W. (in press) The reactivity of Fe(III) (hydr)oxides; effects of ligands in inhibiting the dissolution. Colloids and Surf.
Borghi, E.B., Regazzoni, A.E., Maroto, A.J.G., Blesa, M.A., 1988. Reductive dissolution of magnetite by solutions containing EDTA and FeII. J. Colloid Interface Sci. 130:299–310.
Charlet, L., Manceau, A., 1993. Structure, formation and reactivity of hydrous oxide particles; insights from X-ray absorption spectroscopy. In Environmental Particles (vol. 2) (ed. J. Buffle and H.P. Von Leeuwen) Lewis publ., Boca Raton, Florida.
Dos Santos Afonso, M., Stumm, W., 1992. Reductive dissolution of iron(III) (hydr)oxides by hydrogen sulfide. Langmuir, 8:1671–1675.
Dzombak, D., Morel, F., 1990. Surface Complexation Modeling: Hydrous Ferric Oxide. Wiley-Interscience, New York.
Eggleston, C.M. and W. Stumm, 1993. Scanning tunneling microscopy of Cr(III) chemisorbed onα-Fe2O3 (001) surfaces from aqueous solution: Direct observation of surface mobility and clustering. Geochim. Cosmochim. Acta 57:4843–4850.
Furrer, G., Stumm, W., 1986. The coordination chemistry of weathering: I. Dissolution kinetics ofδ-Al2O3 and BeO. Geochim. Cosmochim. Acta 50:1847–1860.
Pulver, K., Schindler, P.W., Westall, J.C., Grauer, R., 1984. Kinetics and mechanisms of dissolution of Bayerite (γ-Al(OH)3 — HF solutions at 298.2°K, J. Colloid and Interf. Science 10:554–564.
Schindler, P.W., Gamsjäger, H., 1972. Acid-base reactions of the TiO2 (Anatase)-water interface and the point of zero charge of TiO2 suspensions. Kolloid-Z. u. Z-Polymere, 250:759–763.
Schindler, P.W., Kamber, H.R., 1968. Die Acidität von Silanolgruppen, Helv. Chim. Acta 51:1781–1786.
Schindler, P., Stumm, W., 1987. The surface chemistry of oxides, hydroxides, and oxide minerals. In Aquatic Surface Chemistry, (ed. W. Stumm) pp. 83–110. Wiley-Interscience, New York.
Sigg, L., Stumm, W., 1981. The interactions of anions and weak acids with hydrous goethite (α-FeOOH) surface. Colloids and Surfaces 2:101–117.
Sposito, G., 1984. The Surface Chemistry of Soils. Oxford Univ. Press.
Stumm, W., 1992. Chemistry of the Solid — Water Interface, Wiley-Interscience, New York.
Westall, J., 1979. MICROQL — I: A chemical equilibrium program in BASIC, EAWAG, Dübendorf, Switzerland.
Wieland, E., Wehrli, B., Stumm, W., 1988. The coordination chemistry of weathering: III A generalization on the dissolution rates of minerals. Geochim. Cosmochim. Acta: 48, 1969–1981.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Stumm, W. From surface acidity to surface reactivity; inhibition of oxide dissolution. Aquatic Science 55, 273–280 (1993). https://doi.org/10.1007/BF00877272
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00877272