Summary
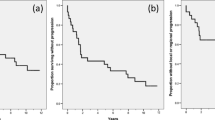
Twenty-two patients with recurrent or metastatic squamous cell carcinoma of the head and neck were treated with piroxantrone 150 mg/m2 intravenously every 21 days. There were no objective responses. The 95% upper confidence bound for response is 15%. Primary toxicity was hematologic.
Similar content being viewed by others
References
Showalter H, Johnson J, Werbel L,et al: 5-[(amino-alkyl)amino] substitutional anthra [1,9-cd] pyrazol-6(2H)-ones as novel anticancer agents. Synthesis and biological evaluations. J Med Chem 27:253–255, 1984
Amus M, Loprinzi C, Collins JM,et al: Phase I and clinical pharmacological evaluation of pirozantrone hydrochloride (oxantrazole). Cancer Res 50:3906–3909, 1990
SWOG 8514 Randomized comparison of cisplatin plus 5-FU vs. CBDO plus F-FU vs. methotrexate in advanced squamous cell carcinomas of the head and neck: A Southwest Oncology Group Study. AA Forastiere, B Metch, DE Schuller, JF Ensley, LF Hutchins, SP Bulcerzak, JA Kish, S McClure, E Von Feldt, SK Williamson, DD Von Hoff. JCO.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Taylor, S.A., Benedetti, J., Schuller, D. et al. Phase II trial of piroxantrone in patients with recurrent and metastatic squamous cell carcinoma of the head and neck. Invest New Drugs 11, 227–229 (1993). https://doi.org/10.1007/BF00874161
Issue Date:
DOI: https://doi.org/10.1007/BF00874161