Summary
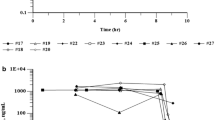
Fourteen patients with advanced renal cell carcinoma were treated on a phase II trial with liposomal encapsulated doxorubicin (Lipodox, LED). None of the fourteen evaluable patients achieved a complete or partial response. Myelosuppression was the most common toxicity and no cardiac toxicity was evident. Seventynine percent (11 of 14) of patients experienced grade III or IV neutropenia. In summary, LED did not show antitumor activity in the treatment of advanced renal cell carcinoma.
Similar content being viewed by others
References
Fojo AT, Shen D-W, Mickley LA, Pastan I, Gottesman MM: Intrinsic drug resistance in human kidney cancer is associated with expression of a human multidrug-resistance gene. J Clin Oncol 5:1922–1927, 1987
Thierry AR, Vige D, Dritschilo A, Rahman A: Modulation of multidrug resistance by liposomally encapsulated doxorubicin in two human MDS tumor cell lines. Proc Am Assoc Cancer Res 32:373, 1991
Oudard S, Thierry A, Jorgensen TJ, Rahman A: Sensitization of multidrug resistant colon cancer cells to doxorubicin encapsulated in liposomes. Cancer Chemother Pharmacol 28:259–265, 1991
Miller AB, Hoogstraten B, Staquet M, Winkler A: Reporting results of cancer treatment. Cancer 47:207–214, 1981
Rahman A, Treat J, Roh J-K, Potkul LA, Alvord WG, Forst D, Woolley PV: A phase I clinical trial and pharmacokinetic evaluation of liposome-encapsulated doxorubicin. J Clin Oncol 8:1093–1100, 1990
Treat J, Greenspan A, Forst D, Sanchez JA, Ferrans VJ, Potkul LA, Woolley PV, Rahman R: Antitumor activity of liposome-encapsulated doxorubicin in advanced breast cancer: phase II study. J Natl Cancer Inst 82:1706–1710, 1990
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Law, T.M., Mencel, P. & Motzer, R.J. Phase II trial of liposomal encapsulated doxorubicin in patients with advanced renal cell carcinoma. Invest New Drugs 12, 323–325 (1994). https://doi.org/10.1007/BF00873048
Issue Date:
DOI: https://doi.org/10.1007/BF00873048