Abstract
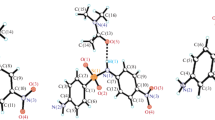
An x-ray structural investigation of crystals of the black modification of 2-(para-dimethylaminophenyl)indan-1,3-dione was carried out. It was found that the molecules in the crystal, which was shown to be the hemihydrate, coexist in the enolic and zwitterion forms. The absence of strong delocalization of the negative charge was shown for the bipolar form. In the crystal, the molecules are bound mutually and with the solvate molecular of water with a system of hydrogen bonds allowing the possible “cooperative” transfer of protons.
Similar content being viewed by others
References
O. Ya. Neiland and V. Ya. Kroge,Izv. Akad. Nauk Latv. SSR, Ser. Khim., No. 4, 483 (1964).
O. Ya. Neiland and Ya. Ya. Linaberg,Izv. Akad. Nauk Latv.SSR, Ser. Khim., No. 6, 691 (1964).
Z. G. Aliev, L. O. Atovmyan, A. M. Mikhailova, L. M. Pisarenko, and V. I. Nikulin,Zh. Strukt. Khim. 30, No. 6, 164 (1989).
A. A. Kemme, M. F. Bundule, Ya. Ya. Bleidelis, É. É. Liepin'sh, É. S. Lavrinovich, and Yu. É. Fridmanis,Khim. Geterotsikl. Soedin., No. 8, 1076 (1978).
V. F. Kaminskii, R. P. Shibaeva, and O. Ya. Neiland,Zh. Strukt. Khim. 17, No. 5, 898 (1976).
V. A. Strel'nikov and V. E. Zavodnik,Kristallografiya 34, No. 6, 1369 (1990).
Additional information
N. N. Semenov Institute of Chemical Physics, Russian Academy of Sciences, 117977 Moscow. A. N. Nesmeyanov Institute of Heteroorganic Compounds, Russian Academy of Sciences, 117813 Moscow. Translated from Izvestiya Akademii Nauk, Seriya Khimicheskaya, No. 9, pp. 2117–2122, September, 1992.
Rights and permissions
About this article
Cite this article
Motyakin, M.V., Yufit, D.S., Pisarenko, L.M. et al. Crystalline and molecular structures of the enol from of 2-(para-dimethylaminophenyl)indan-1,3-dione. Russ Chem Bull 41, 1652–1656 (1992). https://doi.org/10.1007/BF00863588
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00863588