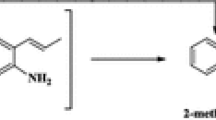Abstract
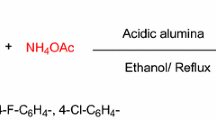
An investigation has been conducted into the staged synthesis of 2-methylimidazole from ethylenediamine and acetic acid in the presence of a bifunctional aluminoplatinum catalyst. It has been shown that the formation stage of 2-methylimidazoline occurs more quickly on γ-Al2O3 than its dehydrogenation on the Pt centers. From a comparison of the processes of dehydrogenation of 2-methylimidazoline in the molten phase and in aqueous solution it follows that the water eliminated in the imidazoline formation stage could cause the decrease in activity during the dehydrogenation on the Pt centers. The structures of the secondary and intermediate products have been established for each stage of the process and their formation routes are discussed.
Similar content being viewed by others
References
US Patent No. 2,847,417;Chem. Abstr.,53, 4305h (1959).
US Patent No. 1,231,249;Chem. Abstr.,64, 2094p (1966).
US Patent No. 1,952,991;Chem. Abstr.,73, 25471Z (1970).
US Patent No. 2,729,017;Chem. Abstr.,90, 186947a (1979).
USSR Inventor's Certificate No. 1,397,440;Byull. Izobret., No. 19 (1988).
K. M. Gitis, G. E. Neumoeva, N. I. Raevskaya, G. A. Arutyunyants, and G. V. Isagulyants,Izv. Akad. Nauk, Ser. Khim., No. 1, 932 (1992).
K. M. Gitis, N. I. Raevskaya, A. G. Zaitsev, V. Yu. Borovkov, S. B. Kogan, and G. V. Isagulyants,Izv. Akad. Nauk Ser. Khim., No. 9, 1992 (1992).
USSR Inventor's Certificate No. 180,605;Byull. Izobret., No. 21 (1967).
K. Hofmann, in:Chemistry of Heterocyclic Compounds, A. Weissberger (ed.), Interscience, New York—London (1959), p. 219.
Additional information
N. D. Zelinskii Institute of Organic Chemistry, Russian Academy of Sciences, 117913 Moscow. Translated from Izvestiya Akademii Nauk, Seriya Khimicheskaya, No. 9, pp. 1997–2002, September, 1992.
Rights and permissions
About this article
Cite this article
Gitis, K.M., Raevskaya, N.I. & Isagulyants, G.V. The staged synthesis of 2-methylimidazole from ethylenediamine and acetic acid in the presence of a bifunctional aluminoplatinum catalyst. Russ Chem Bull 41, 1551–1554 (1992). https://doi.org/10.1007/BF00863570
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00863570