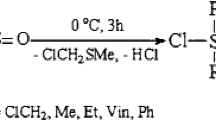Summary
-
1.
The introduction of polar groups in the organic group of organosilanes greatly activates them in thetelomerization reaction with hexamethylcyclotrisiloxane.
-
2.
The activity of (substituted-alkyl)chlorosilanes depends on the position of the polar group relative to the silicon atom and falls in the series β>α >γ.
-
3.
The cyano group has a greater effect than a chlorine atom: CN>Cl.
-
4.
Ten previously undescribed compounds were synthesized.
Similar content being viewed by others
Literature cited
K. A. Andrianov, V. V. Severnyi and B. A. Izmailov, Izv. AN SSSR. Otd. khim. n., 282 (1963).
K. A. Andrianov and V. V. Severnyi, Izv. AN SSSR. Otd. khim. n., 82 (1963).
K. A. Andrianov and V. V. Severnyi, Papers Read at the Twelth Conference on Macromolecular Compounds [in Russian], Baku, (1962).
E. A. Chernyshev, V. F. Mironov and A. D. Petrov, Chemistry and Practical Application of Organosilicon Compounds [in Russian], No. 1, Leningrad (1958), p. 115.
K. A. Andrianov and V. V. Severnyi, Izv. AN SSSR. Ser. khim., 1268 (1964).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 7, pp. 1271–1275, July, 1964
Rights and permissions
About this article
Cite this article
Andrianov, K.A., Severnyi, V.V. Telomerization of dimethylcyclosiloxanes. Communication 7. Preparation of dimethylsiloxane oligomers with functional groups in the organic group. Russ Chem Bull 13, 1178–1181 (1964). https://doi.org/10.1007/BF00863125
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00863125