Abstract
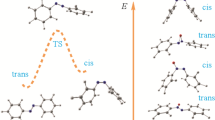
The AM-1 method with full optimization of geometry was used to study several proposed isomerization mechanisms for dichloro- and difluorobenzenes on active centers as proton donors. Preferred positions of proton addition in dihalobenzenes were studied and protonation and activation energies for intramolecular migration of hydrogen and halogen atoms were determined. It was shown that the isomerization mechanisms are the same for dichloro-and difluorobenzenes and result from a 1,2-shift of the halogen. The activation energy for migration of a fluorine atom in difluorobenzenonium cations is substantially higher than for a chlorine atom in dichlorobenzenonium cations. The data obtained on the mechanism have experimental verification.
Similar content being viewed by others
Literature cited
J. Perdillos, D. Brunel, B. Coq, P. Massiani, L. C. De Menorval, and F. Figueras,J. Am. Chem. Soc.,112, No. 4, 1313 (1990).
L. T. Scott and J. S. Highsmith,Tetrahedron Lett.,21, No. 49, 4703 (1980).
M. J. S. Dewar, E. G. Zoebisch, E. F. Healy, and J. J. P. Stewart,J. Am. Chem. Soc.,107, No. 13, 3902 (1985).
J. B. Pedley, R. D. Naylor, and S. P. Kirby (eds.),Thermochemical Data of Organic Compounds, Chapman and Hall, New York (1986).
V. A. Koptyug,Arenonium Ions. Structure and Reactivity [in Russian], Nauka, Novosibirsk (1983).
W. J. Hehre and P. C. Hilberty,J. Am. Chem. Soc.,96, No. 22, 7163 (1974).
F. Cacace and M. Speranza,J. Am. Chem. Soc.,98, No. 23, 7305 (1976).
Additional information
A. N. Nesmeyanov Institute Heteroorganic Compounds, Russian Academy of Sciences, 117813 Moscow. N. D. Zelinskii Institute of Organic Chemistry, Russian Academy of Sciences, 117433 Moscow. Translated from Izvestiya Akademii Nauk, Seriya Khimicheskaya, No. 3, pp. 731–737, March, 1992.
Rights and permissions
About this article
Cite this article
Borisov, Y.A., Raevskii, N.I., Mortikov, E.S. et al. Isomerization of dichloro- and difluorobenzenes: A quantum-chemical consideration. Russ Chem Bull 41, 574–578 (1992). https://doi.org/10.1007/BF00863088
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00863088