Conclusions
-
1.
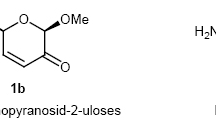
The acetonation ofβ-methyl-D-glycero-D-gulo-heptopyranoside gave the 2, 3∶6, 7-di-O-isopro-pylidene derivative, the oxidation of which gives the 4-keto-heptoside.
-
2.
The obtained ketone was used for the stereodirectional insertion of hydroxy and amino groups into the heptosulose molecule, and also for branching the carbon chain.
Similar content being viewed by others
Literature cited
O. Lüderitz, M. A. Staub, and O. Westphal, Bacteriol. Rev.,30, 192 (1966).
N. K. Kochetkov and B. A. Dmitriev, Tetrahedron,21, 803 (1965).
G. Bagdian, W. Dröge, K. Kotelko, O. Lüderitz, and O. Westphal, Biochem. Z.,344, 197 (1966).
M. J. Osborn, Proc. Natl. Acad. Sci. U. S.,50, 499 (1963).
G. A. Adams, C. Quadling, and M. B. Perry, Can. J. Microbiol.,13, 1605 (1967).
N. J. Antia and M. B. Perry, Can. J. Chem.,38, 1917 (1960).
M. E. Evans, F. W. Parrish, and L. Long, J., Carbohydrate Res.,3, 453 (1967).
D. C. De Jongh and K. Biemann, J. Am. Chem. Soc.,86, 67 (1964).
J. W. Pratt and N. K. Richtmyer, J. Am. Chem. Soc.,77, 6326 (1955).
P. M. Collins and W. G. Overend, J. Chem. Soc., 3448 (1965).
E. J. Corey and M. Chaykovsky, J. Am. Chem. Soc.,87, 1353 (1965).
J. H. Jordan and S. Smedley, Carbohydrate Res.,16, 177 (1971).
H. Agahigian, G. D. Vickers, M. H. von Saltza, J. Reid, A. I. Cohen, and H. Gauthier, J. Org, Chem.,30, 1085 (1965).
B. Coxon, Tetrahedron,22, 2287 (1966).
H. S. Isbell and H. L. Frush, J. Res. Natl. Bur. Std.,24, 125 (1940).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 10, pp. 2298–2304, October, 1972.
Rights and permissions
About this article
Cite this article
Dmitriev, B.A., Chernyak, A.Y., Chizhov, O.S. et al. Monosaccharides. Russ Chem Bull 21, 2230–2235 (1972). https://doi.org/10.1007/BF00855304
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00855304