Conclusions
-
1.
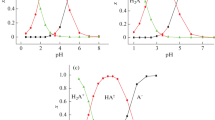
The dependence of the position of the equilibrium between the meso- and racemic forms of diaminopimelic acid (DPA) on the pH was studied, and their dissociation constants at 22 and 60° were determined.
-
2.
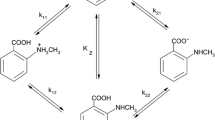
The particular equilibrium constants of differently ionized forms of DPA were determined, and it was shown that for the bis-protonated bianion, the racemic form of DPA is more stable, for the monoprotonated form the meso-form, and for the bianion the stability of the two isomers is the same.
-
3.
The observable differences in the values of the particular constants are explained by intramolecular interaction of the charged groups in the DPA molecule.
Similar content being viewed by others
Literature cited
M. Antia, D. Hoare, and E. Work, Biochem. J.,56, 448 (1957).
P. White and B. Kelly, Biochem. J.,96, 75 (1965).
B. Gorton, J. Coker, and H. Browder, Indust. and Engng. Chem.,2 (4), 313 (1963).
G. A. Kotova and V. M. Belikov, Izv. Akad. Nauk SSSR, Ser. Khim., 1163 (1970).
K. Hirota and Y. Izumi, Bull. Chem. Soc. Japan,40, 178 (1967).
G. A. Kotova and V. M. Belikov, Mikrobiol. Prom-St',7, 35 (1970).
R. Wade, S. Birnbaum, M. Winitz, and J. Greenstein, J. Amer. Chem. Soc.,79, 648 (1957).
A. Albert and E. Serjeant, Ionization Constants of Acids and Bases [Russian translation], Khimiya, Moscow-Leningrad (1964).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 9, pp. 1951–1955, September, 1972.
Rights and permissions
About this article
Cite this article
Belikov, V.M., Kotova, G.A. & Belokon', Y.N. Configurational equilibrium ofα,α′-diaminopimelic acid in aqueous solution at various pH. Russ Chem Bull 21, 1894–1897 (1972). https://doi.org/10.1007/BF00854600
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00854600