Conclusions
-
1.
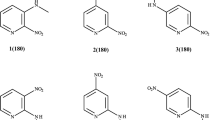
Based on the data of the NMR spectra, primary and secondary 4-amino-3-acetyl-6-methyl-2-pyrones (IV) exist predominantly in tautomeric form (IVA), which contains a stable intramolecular hydrogen bond.
-
2.
The chemical shifts of the NH proton in a ring of the chelate type depend importantly on the nature of the substituent attached to the nitrogen atom, and reach their highest value for compounds containing an aromatic substituent.
-
3.
In the primaryγ-aminopyrone the chemical shifts of both NH protons differ by ~ 1.6 ppm, while the JN 15,H spin-spin coupling constants differ by 1 Hz.
Similar content being viewed by others
Literature cited
F. M. Dean, Naturally Occurring Oxygen Ring Compounds, Butterworth and Co. Ltd., London (1963), Chap. 4.
S. Forsén and M. Nilsson, Arkiv Kemi,17, 523 (1961).
S. Garrat, J. Org. Chem.,28, 1886 (1963).
R. N. Schut, W. G. Strycker, and T. M. H. Liu, J. Org. Chem.,28, 3046 (1963).
J. D. Edwards, J. E. Page, and M. Pianka, J. Chem. Soc., 5200 (1964).
A. A. Akhrem, A. M. Moiseenkov, F. A. Lakhvich, V. I. Kadentsev, and O. S. Chizhov, Izv. Akad. Nauk SSSR, Ser. Khim., 1206 (1970).
A. A. Akhrem, A. M. Moiseenkov, F. A. Lakhvich, S. P. Smul'skii, and V. A. Petukhov, Izv. Akad. Nauk SSSR, Ser. Khim., 917 (1972).
A. A. Akhrem, A. M. Moiseenkov, F. A. Lakhvich, and A. I. Poselenov, Izv. Akad. Nauk SSSR, Ser. Khim., 143 (1972).
G. Binsch, J. B. Lambert, B. W. Roberts, and J. D. Roberts, J. Am. Chem. Soc.,86, 5564 (1964).
A. A. Akhrem, A. M. Moiseenkov, A. I. Poselenov, V. A. Petukhov, and V. S. Bogdanov, Izv. Akad. Nauk SSSR, Ser. Khim., 1746 (1971).
V. S. Bogdanov, V. V. Negrebetskii, V. A. Korenevskii, A. M. Moiseenkov, F. A. Lakhvich, and A. A. Akhrem, Izv. Akad. Nauk SSSR, Ser. Khim., 550 (1971).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 5, pp. 1114–1118, May, 1972.
Rights and permissions
About this article
Cite this article
Bogdanov, V.S., Negrebetskii, V.V., Lakhvich, F.A. et al. Study of the structure of 4-amino-3-acetyl-6-methyl-2-pyrones by NMR. Russ Chem Bull 21, 1067–1070 (1972). https://doi.org/10.1007/BF00853770
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00853770