Conclusions
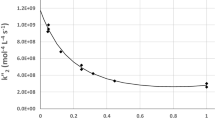
The rate constants of reactions of atomic oxygen with methane, ethane, ethylene, and propylene along different pathways were determined on the basis of the ratios between the concentrations of the products formed and the summary rate constants measured earlier.
Similar content being viewed by others
Literature cited
L I. Avramenko, R. V. Kolesnikova, and N. L. Kuznetsova, Izv. Akad. Nauk SSSR, Otd. Khim. Nauk, 620 (1963).
L. I. Avramenko, R. V. Kolesnikova, and G. I. Savinova, Izv. Akad. Nauk SSSR, Otd. Khim. Nauk, 976 (1963).
L. I. Avramenko, R. V. Kolesnikova, and G. I. Savinova, Izv. Akad. Nauk SSSR, Otd. Khim. Nauk, 36 (1963); Ser. Khim., 253 (1967).
R. L. Pusell and F. S. Rowland, J. Amer. Chem. Soc.,90, 1671 (1968).
E. L. Wong and A. E. Potter, J. Chem. Phys.,39, 2211 (1963).
V. V. Azatyan and A. B. Nalbandyan, Zh. Vses. Khim. O-va im. Mendeleeva,11, 162 (1966).
R. D. Cadle and E. R. Allen, J. Phys. Chem.,69, 1611 (1965).
L. I. Avramenko, R. V. Kolesnikova, and G. I. Savinova, Izv. Akad. Nauk SSSR, Ser. Khim., 408 (1965).
L. I. Avramenko and R. V. Kolesnikova, Problems of Chemical Kinetics, Catalysis, and Reactivity [in Russian], Izd-vo AN SSSR (1956), p. 7.
N. N. Semenov, Some Problems of Chemical Kinetics and Reactivity [in Russian], Izd-vo AN SSSR (1958).
R. J. Cvetanovic, Canad. J. Chem.,36, 623 (1958).
L. I. Avramenko and R. V. Kolesnikova, Izv. Akad. Nauk SSSR, Ser. Khim., 327 (1970).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 12, pp. 2693–2699, December, 1971.
Rights and permissions
About this article
Cite this article
Avramenko, L.I., Kolesnikova, R.V. Determination of the elementary reaction rate constants on the basis of the summary values of the rate constants. Russ Chem Bull 20, 2556–2561 (1971). https://doi.org/10.1007/BF00853622
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00853622