Conclusions
-
1.
The conditions of synthesis and isolation of crystal hydrates of copper and nickel nitrilotriacetates were determined. Previously undescribed nitrilotriacetates with the composition HNiX·3H2O, KH(CuX)2·3H2O, Ni2X3·9H2O were isolated, where X3− is the acid residue of nitrilotriacetic acid.
-
2.
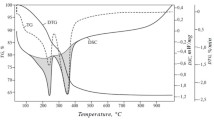
The content of water of hydration in the synthesized solid nitrilotriacetates was determined. pH-metric, thermogravimetric, and x-ray diffraction investigations of copper and nickel nitriloacetates were conducted.
Similar content being viewed by others
Literature cited
G. Schwarzenback, A. Willi, and R. Bach, Helv. Chim. Acta,30, 1303 (1947).
B. Kirson and R. Bornstin, Bull. Soc. Chim. France,6, 1081 (1961).
S. Yu. Elovich and L. G. Tonkonog, Zh. Neorgan. Khimii,6, 1791 (1961).
J. V. Dubski and M. Spritzmann, J. Pract. Chem.,96, 112 (1917).
J. Tomita and K. Ueno, Bull. Chem. Soc., Japan,36, No. 9, 1069 (1963).
H. Brintzinger and G. Hesse, Z. Anorgan. und Allgem. Chem.,249, 299 (1942).
P. Pfeiffer and W. Offermann, Ber.,75, No. 1 (1942).
W. Kelmm and K. H. Raddatz, Z. Anorgan. und Allgem. Chem.,250, 556 (1959).
G. Schwarzenbach and R. Prshibil, Complexometry [Russian translation], GNTIKhL, Moscow (1958).
G. S. Smith and J. L. Hoard, J. Amer. Chem. Soc.,81, 556 (1959).
L. I. Martynenko, N. I. Pechurova, E. N. Efremov, and A. I. Grigor'ev, Zh. Neorgan. Khimii,12, 424 (1967).
L. I. Martynenko, N. I. Pechurova, Vikt. I. Spitsyn, Ali Khadi, K. I. Popov, and Nguen Dun' Hgo, Izv. Akad. Nauk SSSR, Ser. Khim., 2659 (1970).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 10, pp. 2115–2121, October, 1971.
Rights and permissions
About this article
Cite this article
Mitrofanova, N.D., Martynenko, L.I. & Spitsyn, V.I. Comparative investigation of solid copper and nickel nitrilotriacetates. Russ Chem Bull 20, 2005–2009 (1971). https://doi.org/10.1007/BF00851238
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00851238