Conclusions
-
1.
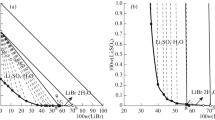
The liquid-solid equilibrium in the system LiAlH4-diglyme was studied in the temperature interval from −65 to 70°, and that in the system LiAlH4-KAlH4-diglyme at 25°.
-
2.
The existence of the solvates LiAlH4·3DG and LiAlH4·2DG was established.
-
3.
LiAlH4, KAlH4, and diglyme form a simple eutonic system; the solubility isotherm at 25° consists of the branches of crystallization of LiAlH4·2DG and KAlH4 with eutonic composition 11.8 mole % LiAlH4, 17.4 mole % KAlH4, and 70.8 mole % diglyme.
Similar content being viewed by others
Literature cited
H. Clasen, Angew. Chemie,73, 46 (1961).
S. I. Bakum and T. N. Dymova, Izv. Akad. Nauk SSSR, Ser. Khim., 1890 (1970).
T. N. Dymova, S. I. Bakum, S. S. Grazhulene, and M. Mukhidinov, Izv. Akad. Nauk SSSR, Ser. Khim., 1892 (1970).
S. I. Bakum, U. Mirsaidov, M. E. Kost, and T. N. Dymova, Izv. Akad. Nauk SSSR, Ser. Khim., 2108 (1972).
G. A. Osipov, M. S. Belyaeva, G. K. Klimenko, L. I. Zakharkin, and V. V. Gavrilenko, Kinetika i Kataliz, 11, 901 (1970).
R. Prshibil, Complexones in Chemical Analysis [Russian translation], IL (1960).
N. S. Poluektov, Methods of Analysis According to Flame Photometry [in Russian], Khimiya (1967).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No.2, pp.259–261, February, 1973.
Rights and permissions
About this article
Cite this article
Mirsaidov, U., Bakum, S.I. & Dymova, T.N. Solubility diagram of the system LiAlH4-diglyme and solubility isotherm in the system LiAlH4-KAlH4-diglyme at 25°. Russ Chem Bull 22, 251–253 (1973). https://doi.org/10.1007/BF00850966
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00850966