Conclusions
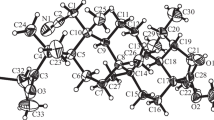
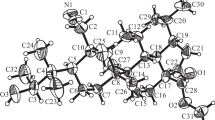
In the cyclization of 3-methoxy-8, 14-seco-D-homoestra-1,3,5,(10),9(11)-tetraene-14, 17a-dione (I) the stereoisomeric 14α-hydroxy-3-methoxy-D-homoestra-1,3,5(10),9(11)-tetraen-17a-ones (III) and (IV) are formed at first, and after dehydration and isomerization these are converted into 3-methoxy-D-homoestra-1,3,5(10),8,14-pentaen-17a-one (II). The ketol (IV) is much more readily dehydrated than the ketol (III) and is probably themain intermediate product in the cyclization of the diketone (I).
Similar content being viewed by others
Literature cited
S. N. Ananchenko and I. V. Torgov, Dokl. AN SSSR.127, 553 (1959).
S. N. Ananchenko, V. N. Leonov, A. V. Platonova, and I. V. Torgov, Dokl. AN SSSR135, 73 (1960).
P. A. Robins and J. Walker, J. Chem. Soc. 3249, 3260 (1956).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akadeemii Nauk SSSR, Seriya Khimicheskaya, No. 8, pp. 1413–1416, August, 1965 Original article submitted June 24, 1963
Rights and permissions
About this article
Cite this article
Zakharychev, A.V., Ananchenko, S.N. & Torgov, I.V. Cyclization of 3-methoxy-8, 14-seco-D-homoestra-1, 3, 5(10), 9(11)-tetraene-14, 17a-dione into D-homoestrone derivatives. Russ Chem Bull 14, 1373–1375 (1965). https://doi.org/10.1007/BF00846195
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00846195