Summary
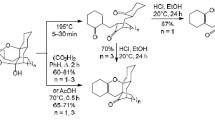
The stereochemistry of the bromination of 4-methyl-cis-4-cyclohexene-l,2-dicarboxylic acid (I) and its anhydride (II) was studied. The spatial structures of the products were proved, and a possible mechanism scheme for their formation was discussed.
Similar content being viewed by others
Literature cited
V. F. Kucherov, A. L. Shabanov, and A. S. Onishchenko, Izv. AN SSSR, Otd. Khim. n., 1963, 844.
A. S. Onishchenko, A. L. Shabanov, and V. F. Kucherov, Izv. AN SSSR, Otd. khim. n., 1963, 852.
K. Crewe, A. Heinke, and Ch. Sommer, Chem. Ber.,89, 1978 (1956).
A. L. Shabanov, A. S. Onishchenko, and V. F. Kucherov, Izv. AN SSSR, Otd. khim. n., 1963, 1790.
I. N. Nazarov, V. F. Kucherov, and V. M. Andreev, Izv. AN SSSR, Otd. khim. n., 1955, 289; 1956, 951; 1959, 1058.
Author information
Authors and Affiliations
Additional information
This article is published in accordance with a resolution of the Conference of Chief Editors of the Journals of the Academy of Sciences of the USSR, July 12, 1962, as a dissertation paper by A. L. Shabanov.
Rights and permissions
About this article
Cite this article
Shabanov, A.L., Onishchenko, A.S. & Kucherov, V.F. Stereochemistry of cyclic compounds Communication 63. Stereochemistry of the bromination of 4-methyl-cis-4-cyclohexene-1,2-dicarboxylic acid and its anhydride. Russ Chem Bull 12, 1648–1652 (1963). https://doi.org/10.1007/BF00845794
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00845794