Summary
-
1.
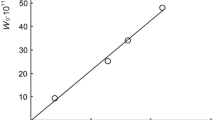
The inhibitor method has been used to make a study of the decay into free radicals of α-ketocyclohexyl-hydroperoxide, formed in the oxidation of cyclohexanone. In cyclohexanone, the peroxide decays monomolecularly with a rate constant equal to 3·107 e−20400/RT sec−1.
-
2.
In cyclohexanone, the α-ketocyclohexylhydroperoxide joins reversibly to the ketone to form a peroxide which rapidly decays into free radicals.
Similar content being viewed by others
Literature cited
I. V. Berezin, E. T. Denisov, and N. M. Émanuél', Collection: Problems in Chemical Kinetics, Catalysis, and Reactivity [in Russian], Academy of Sciences Press, USSR, p. 273 (1955).
E. T. Denisov and V. V. Kharitonov, Zh. fiz. khimii,35, 444 (1961).
I. V. Berezin, N. F. Kazanskaya, and V. F. Privalov, Dokl. AN SSSR,126, 809 (1959).
E. T. Denisov, Zh. Fiz. khimii,33, 1198 (1959).
W. Pritzkow, Ber.,87, 1668 (1954).
E. T. Denisov and N. M. Émanuél', Ustekhi akhimii,27, 365 (1958).
A. Farkas and E. Pasagla, J. Amer. Chem. Soc.,72, 3333 (1950).
Z. K. Maizus, I. P. Skibida, and N. M. Émanuél', Dokl. AN SSSR,131, 880 (1960).
E. T. Denisov, Dokl. AN SSSR,146, 394 (1962).
G. I. Zaikov, Z. K. Maizus, and N. M. Émanuél', Dokl. AN SSSR,140, 405 (1961).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Denisov, E.T., Denisova, L.N. Degenerate chain branching mechanism in cyclohexanone undergoing oxidation. Russ Chem Bull 12, 1589–1593 (1963). https://doi.org/10.1007/BF00845784
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00845784