Conclusions
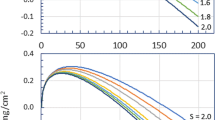
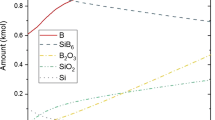
The kinetics of oxidation of the Ni3Ti compound at temperatures from 650 to 1000 °C follows, essentially, a parabolic time function. The mechanism of this process changes with temperature and exposure time, as is indicated by a deviation from the parabolic mode of oxidation, by the irregularities in the time and the temperature characteristics of oxidizability and of the parabolic rate “constant,” and by the change in the external appearance of the complexly constituted scale.
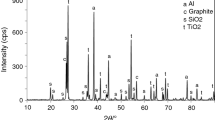
An intensive formation of TiO2 grains results in a linear oxidation mode at t ∼ 800 °C and a much greater oxidizability at temperatures above 800 °C.
Similar content being viewed by others
Literature cited
I. I. Kornilov, Izv. Akad. Nauk SSSR, Metallurgiya i Toplivo, No. 6 (1966).
P. Duwez and J. L. Taylor, Trans. Amer. Inst. Mining and Metal. Engr.,188, 1173 (1950); J. Metals,2, No. 9, 1173 (1950).
M. P. Arbuzov and V. G. Chuprina, Izv. Vuzov. SSSR, Fizika, No. 1, 93 (1964).
O. Kubashevski and B. Hopkins, Oxidation of Metals and Alloys [Russian translation], Izd. Inostr. Lit., Moscow (1955).
K. Hauffe, Reactions in Solids and on Their Surfaces [Russian translation], Izd. Inostr. Lit., Moscow (1963).
A. V. Revyakin, in: Titanium and Its Alloys; Metallurgy of Titanium [in Russian], Izd. Akad. Nauk SSSR, Moscow (1962), No. 8, p. 175.
Z. I. Kornilova and D. V. Ignatov, Recent Studies of Titanium Alloys [in Russian], Izd. Nauka, Moscow (1965), p. 143.
A. V. Guts and A. S. Tumarev, Trudy Leningradsk. Politekh. Inst., No. 251, 70 (1965).
M. Davis and C. Birchenall, J. Metals,3, 877 (1951).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya VUZ. Fizika, No. 12, pp. 92–96, December, 1971.
Rights and permissions
About this article
Cite this article
Arbuzov, M.P., Chuprina, V.G. A study conceening the kinetics of oxidation of the Ni3Ti compound. Soviet Physics Journal 14, 1681–1684 (1971). https://doi.org/10.1007/BF00819606
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00819606