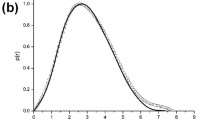
Summary
Crystalline tubular aggregates of actin spontaneously assemble in the presence of certain of the lanthanide ions. These tubes are now known to contain a high degree of structural order and it has been suggested that they may be sensitive to small changes in the primary sequence. However, there have been no detailed studies of the effects of solution conditions associated with their formation. In this report we systematically examine the effects of lanthanide ion concentration, ionic radius, adenosine nucleotide concentration, divalent cation concentration, pH, KCl concentration and incubation time. The stringent control of these parameters leads to a high degree of predictability of the structural parameters of the tubes and will thus be of use in identifying actin isozymes.
Similar content being viewed by others
References
AEBI, U., SMITH, P. R., ISENBERG, G. & POLLARD, T. D. (1980) Structure of crystalline actin sheets.Nature 288, 296–8.
BARDEN, J. A., CURMI, P. M. G. & DOS REMEDIOS, C. G. (1982) Differences between skeletal muscle actin from rabbit and pig.Micron 13, 379–80.
BARDEN, J. A. & DOS REMEDIOS, C. G. (1978) Evidence for the non-filamentous aggregation of actin induced by lanthanide ions.Biochim. biophys. Acta 537, 417–27.
BARDEN, J. A. & DOS REMEDIOS, C. G. (1979) Binding stoichiometry of gadolinium to actin: its effects on the actin-bound divalent cation.Biochem. biophys. Res. Commun. 86, 529–35.
BARDEN, J. A., TULLOCH, P. A. & DOS REMEDIOS, C. G. (1981) Crystalline actin tubes. IV. Structural information on actin monomers obtained from computer-averaged lattice images.J. Biochem. 90, 287–90.
CARSTEN, M. E. & MOMMAERTS, W. F. H. M. (1963) A study of actin by means of starch gel electrophoresis.Biochemistry 2, 28–34.
CURMI, P. M. G., TULLOCH, P. A., DOS REMEDIOS, C. G. & BARDEN, J. A. (1982) Electron diffraction and computed Fourier transform study of lanthanide actin microcrystals.Micron 13, 383–4.
DOS REMEDIOS, C. G. & BARDEN J. A. (1977) Effects of Gd(III) on G-actin: inhibition of polymerization of G-actin and activation of myosin ATPase activity by Gd-G-actin.Biochem. biophys. Res. Commun. 77, 1339–46.
DOS REMEDIOS, C. G., BARDEN, J. A. & VALOIS, A. A. (1980a) Crystalline actin tubes. II. The effect of various lanthanide ions on actin tube formation.Biochim. biophys. Acta 624, 174–86.
DOS REMEDIOS, C. G., BARDEN, J. A., VALOIS, A. A. & TULLOCH, P. A. (1980b) Actin tube structures formed by Gd3+ and analyzed by electron microscopy and electron diffraction. InFibrous Proteins: Scientific, Industrial and Medical Aspects (edited by PARRY, D. A. D. and CREAMER, L. K.), Vol. 2, pp. 23–32. New York: Academic Press.
DOS REMEDIOS, C. G. & DICKENS, M. J. (1978) Actin microcrystals and tubes formed in the presence of gadolinium ions.Nature 276, 731–3.
FINLAYSON, P. J. & DOS REMEDIOS, C. G. (1981) Difference between cardiac and skeletal muscle actins.J. molec. cell. Cardiol. 13, 1081–6.
KISELEV, N. A., SHPITZBERG, C. L. & VAINSHTEIN, B. K. (1967) Crystallization of catalase in the form of tubes with monomolecular walls.J. molec. Biol. 25, 433–41.
NAKAMURA, T., YAMAGUCHI, M. & YANAGISAWA, T. (1979) Comparative studies of actins from various sources.J. Biochem. 85, 627–31.
OWEN, M. J., AUGER, J., BARBER, E. H., EDWARDS, A. J., WALSH, F. S. & CRUMPTON, M. J. (1978) Actin may be present on the lymphocyte surface.Proc. natn. Acad. Sci. USA 75, 4484–8.
SHANNON, R. D. (1976) Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides.Acta Cryst. A32, 751–67.
SMITH, P. R., FOWLER, W. E., POLLARD, T. D. & AEBI, U. (1983) Structure of the actin molecule determined from electron micrographs of crystalline actin sheets with a tentative alignment of the molecule in the actin filament.J. molec. Biol. 167, 641–60.
SPUDICH, J. A. & WATT, S. (1971) The regulation of rabbit skeletal muscle contraction. I. Biochemical studies of the interaction of the tropomyosin-troponin complex with actin and the proteolytic fragments of myosin.J. biol. Chem. 216, 4866–71.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Curmi, P.M.G., Barden, J.A. & Dos Remedios, C.G. Actin tube formation: effects of variations in commonly used solvent conditions. J Muscle Res Cell Motil 5, 423–430 (1984). https://doi.org/10.1007/BF00818260
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00818260