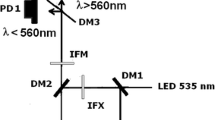
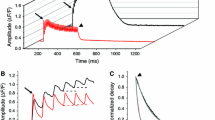
Abstract
Recent studies of isolated muscle membrane have enabled induction and monitoring of rapid Ca2+ release from sarcoplasmic reticulum (SR)5 in vitro by a variety of methods. On the other hand, various proteins that may be directly or indirectly involved in the Ca2+ release mechanism have begun to be unveiled. In this mini-review, we attempt to deduce the molecular mechanism by which Ca2+ release is induced, regulated, and performed, by combining the updated information of the Ca2+ release kinetics with the accumulated knowledge about the key molecular components.
Similar content being viewed by others
References
Aaron, B.-M. B., Oikawa, K., Reithmeier, R. A. F., and Sykes, B. D. (1984).J. Biol. Chem. 259, 11,876–11,881.
Abramson, J. J., Trimm, J. L., Weden, L., and Salama, G. (1983).Proc. Natl. Acad. Sci. USA 80, 1526–1530.
Abramson, J. J., Cronin, J. R., and Salama, G. (1988).Arch. Biochem. Biophys. 263, 245–255.
Antoniu, B., Kim, D. H., Morii, M., and Ikemoto, N. (1985).Biochim. Biophys. Acta 816, 9–17.
Beeler, T., and Gable, K., (1985).Biochim. Biophys. Acta 821, 142–152.
Campbell, K. P., and MacLennan, D. H. (1982).J. Biol. Chem. 257, 1238–1246.
Caswell, A. H., Lau, Y. H., Garcia, M., and Brunshwig, J.-P. (1979).J. Biol. Chem. 254, 202–208.
Chadwick, C. C., Inui, M., and Fleischer, S. (1988).J. Biol. Chem. 263, 10,872–10,877.
Chandler, W. K., Pakowsky, R. F., and Schneider, M. F. (1976).J. Physiol (London.) 254, 285–316.
Chiesi, M., and Wen, Y. S. (1983).J. Biol. Chem. 258, 6078–6085.
Cifuentes, M. E., Hidalgo, C., and Ikemoto, N. (1987).Biophys. J. 51, 351a.
Cifuentes, M. E., Ronjat, M., and Ikemoto, N. (1988).Biophys. J. 53, 131a.
Costello, B., Chadwick, C., Saito, A., Chu, A., Maurer, A., and Fleischer, S. (1986).J. Biol. Chem. 103, 741–753.
Danko, S., Kim, D. H., Sreter, F. A., and Ikemoto, N. (1985).Biochim. Biophys. Acta 816, 18–24.
Donaldson, S. K. B. (1985).J. Gen. Physiol. 86, 501–525.
Donaldson, S. K., Goldblerg, N. D., Walseth, T. F., and Huetteman, D. A. (1987).Biochim. Biophys. Acta 927, 92–99.
Endo, M. (1977).Physiol. Rev. 57, 71–108.
Fabiato, A. (1982).Can. J. Physiol. Pharmacol. 60, 556–567.
Fabiato, A. (1985a).J. Gen. Physiol. 85, 189–246.
Fabiato, A. (1985b).J. Gen. Physiol. 85, 247–289.
Fabiato, A. (1985c).J. Gen. Physiol. 85, 291–320.
Ferguson, D. G., Schwartz, H. W., and Franzini-Armstrong, C. (1984).J. Cell Biol. 99, 1735–1742.
Fleischer, S., Olgunbunmi, E. A., Dixon, M. C., and Fleer, E. A. M. (1985).Proc. Natl. Acad. Sci. USA 82, 7256–7259.
Fliegel, L., Ohnishi, M., Carpenter, M. R., Khanna, V. K., Reithmeier, R. A. F., and MacLennan, D. H. (1987).Proc. Natl. Acad. Sci. USA 84, 1167–1171.
Franzini-Armstrong, C., Kenney, L. J., and Varriano-Marston, E. (1987).J. Cell Biol. 105, 49–56.
Hidalgo, C., and Jaimovich, E. (1989).J. Bioenerg. Biomembr. 21, 267–282.
Hymel, L., Inui, M., Fleischer, S., and Schindler, H. G. (1988).Proc. Natl. Acad. Sci. USA 85, 441–445.
Ikemoto, N., Nagy, B., Bhatnagar, G. M., and Gergely, J. (1974).J. Biol. Chem. 249, 2357–2365.
Ikemoto, N., Antoniu, B., and Kim, D. H. (1984).J. Biol. Chem. 259, 13,151–13,158.
Ikemoto, N., Antoniu, B., and Mészáros, L. G. (1985).J. Biol. Chem. 268, 14,096–14,100.
Ikemoto, N., and Koshita, M. (1988).Biophys. J. 53, 42a.
Ikemoto, N., Ronjat, M., and Mészáros, L. G. (1989).Biophys. J. 55, 14a.
Imagawa, T., Smith, J. S., Coronado, R., and Campbell, K. P. (1987).J. Biol. Chem. 262, 16,636–16,643.
Inesi, G., and Malan, N. (1976).Life Sci. 18, 773–780.
Kasai, M., and Miyamoto, H. (1976a)J. Biochem. 79, 1053–1066.
Kasai, M., and Miyamoto, H. (1976b).J. Biochem. 79, 1067–1076.
Katz, A. M., Repke, D. I., and Hasselbach, W. (1977a).J. Biol. Chem. 252, 1938–1949.
Katz, A. M., Repke, D. I., Dunnett, J., and Hasselbach, W. (1977b).J. Biol. Chem. 252, 1950–1956.
Katz, A. M., Repke, D. I., Fudyma, G., and Shigekawa, M. (1977c).J. Biol. Chem. 252, 4210–4214.
Katz, A. M., Louis, C. F., Repke, D. I., Fudyma, G., Nash-Adler, P. A., Kapsaw, R., and Shigekawa, M. (1980).Biochim. Biophys. Acta 596, 94–107.
Kawamoto, R. M., Brunschwig, J.-P., Kim, K. C., and Caswell, A. H. (1986).J. Cell Biol. 103, 1405–1414.
Kim, D. H., and Ikemoto, N. (1986).J. Biol. Chem. 261, 11,674–11,679.
Kim, D. H., Ohnishi, S. T., and Ikemoto, N. (1983).J. Biol. Chem. 258, 9662–9668.
Kirino, Y., Osakabe, M., and Shimizu, H. (1983).J. Biochem. 94, 1111–1118.
Kometani, T., and Kasai, M. (1978).J. Membr. Biol. 41, 295–308.
Kurebayashi, N., and Ogawa, Y. (1984).J. Biochem. 96, 1249–1255.
Lai, F. A., Erickson, H. P., Rousseau, E., Liu, Q. Y., and Meissner, G. (1988).Nature 331, 315–319.
Lai, F. A., and Meissner, G. (1989).J. Bioenerg. Biomembr. 21, 227–246.
Lattanzio, F. A., Jr., Schlattere, R. C., Nicar, M., Campbell, K. P., and Sutko, J. L. (1987).J. Biol. Chem. 262, 2711–2718.
Martonosi, A. N. (1984).Physiol. Rev. 64, 1240–1320.
Meissner, G. (1984).J. Biol. Chem. 259, 2365–2374.
Meissner, G. (1986).Biochemistry 25, 244–251.
Meissner, G. (1988).Nature 331, 315–319.
Meissner, G., and Henderson, J. S. (1987).J. Biol. Chem. 262, 3065–3073.
Meissner, G., and McKinley, D. (1976).J. Membr. Biol. 30, 79–98.
Meissner, G., Darling, E., and Eveleth, J. (1986).Biochemistry 25, 236–244.
Mészáros, L. G., and Ikemoto, N. (1985a).J. Biol. Chem. 260, 16,076–16,079.
Mészáros, L. G., and Ikemoto, N. (1985b).Biochem. Biophys. Res. Commun. 1127, 836–842.
Mészáros, L. G., Brown, K. L., and Ikemoto, N. (1987).J. Biol. Chem. 262, 11,553–11,558.
Mészáros, L. G., and Ikemoto, N. (1987)Biophys. J. 51, 400a.
Mitchell, R. D., Simmerman, H. K. B., and Jones, L. (1988).J. Biol. Chem. 263, 1376–1381.
Mobley, B. A. (1979).J. Membr. Biol. 46, 315–329.
Morii, H., Takisawa, H., and Yamamoto, T. (1985).J. biol. Chem. 260, 11,536–11,541.
Morii, M., Danko, S., Kim, D. H., and Ikemoto, N. (1986).J. Biol. Chem. 261, 2343–2348.
Moutin, M.-J., and Dupont, Y. (1988).J. Biol. Chem. 263, 4228–4235.
Nagasaki, K., and Kasai, M. (1983).J. Biochem. 94, 1101–1109.
Nagura, S., Kawasaki, T., Taguchi, T., and Kasai, M. (1988).J. Biochem. 104, 461–465.
Nakamura, Y., Kobayashi, J., Gilmore, J., Mascal, M., Rinehart, K. L., Jr., Nakamura, H., and Ohizumi, Y. (1986).261, 4139–4142.
Ogawa, Y., and Kurebayashi, N. (1982).J. Biochem. 92, 899–905.
Ohnishi, S. T. (1979).J. Biochem. 86, 1147–1150.
Ohnishi, S. T. (1987).Biochim. Biophys. Acta 897, 261–268.
Ostwald, T. J., and MacLennan, D. H. (1974).J. Biol. Chem. 249, 974–979.
Palade, P. (1987).J. Biol. Chem. 262, 6142–6148.
Palade, P., Mitchell, R. D., and Fleischer, S. (1983).J. Biol. Chem. 218, 8098–8107.
Rios, E., and Brum, G. (1987).Nature 325, 717–720.
Ronjat, M., and Ikemoto, N. (1989).Biophys. J. 55, 13a.
Rosemblatt, M. S., Cifuentes, M. E., and Ikemoto, N. (1988).Biophys. J. 53, 131a.
Rousseau, E., LaDine, J., Lin, Q.-Y., and Meissner, G. (1988).Arch. Biochem. Biophys. 267, 75–86.
Salama, G., and Abramson, J. (1984).J. Biol. Chem. 259, 13,363–13,369.
Schneider, M. F., and Chandler, W. K. (1973).Nature 242, 244–246.
Seiler, S., Wegener, A. D., Whang, D. D., Hathaway, D. R., and Jones, L. R. (1984).J. Biol. Chem. 259, 8550–8557.
Shoshan, V., MacLennan, D. H., and Wood, D. S. (1981).Proc. Natl. Acad. Sci. USA 78, 4828–4832.
Shoshan-Barmatz, V. (1986).Biochem. J. 240, 509–517.
Smith, J. S., Coronado, R., and Meissner, G. (1985).Nature 316, 446–449.
Stephenson, E. W. (1985).J. Gen. Physiol. 86, 813–832.
Suarez-Isla, B. A., Orozco, C., Heller, P. F., and Froehlich, J. P. (1986).Proc. Natl. Acad. Sci. USA 83, 7741–7745.
Suko, J. (1988).Biochim. Biophys. Acta 938, 79–88.
Sumbillar, C., and Inesi, G. (1987).FEBS Lett. 210, 31–36.
Tanabe, T., Takeshima, H., Mikami, A., Flockerzi, V., Takahashi, H., Kanagawa, K., Kojima, M., Matsuo, H., Hirose, T., and Numa, S. (1987).Nature 328, 313–318.
Vergara, J., Tsien, R. Y., and Delay, M. (1985).Proc. Natl. Acad. Sci. USA 82, 6352–6356.
Volpe, P., and Stephenson, E. W. (1986).J. Gen. Physiol. 87, 271–288.
Volpe, P., Salviati, G., Di Virgilio, F., and Pozzan, T. (1985).Nature 316, 347–349.
Zorzato, F., and Volpe, P. (1988).Biophys. J. 53, 419a.
Zorzato, F., Salviati, G., Facchinett, T., and Volpe, P. (1985).J. Biol. Chem. 260, 7349–7355.
Zorzato, F., Margreth, A., and Volpe, P. (1986).J. Biol. Chem. 261, 13,252–13,257.
Author information
Authors and Affiliations
Additional information
Abbreviations used: AMP-PCP, adenosine 5′-(β, γ-methylenetriphosphate); C1/2, concentration a half-maximal activation or inhibition; Con-A, concanavalin A; DACM,N-(7-dimethylamino-4-methyl-3-coumarinyl)maleimide; DCCD, dicyclohexylcarbodiimide; SR, sarcoplasmic reticulum; DHP, dihydropyridine; E-C, excitation-contraction; EP, phosphorylated intermediate of the enzyme; IP3, inositol 1,4,5-trisphosphate; JFM, junctional face membrane;M r, molecular weight; T-tubule, transverse-tubular system.
Rights and permissions
About this article
Cite this article
Ikemoto, N., Ronjat, M. & Mészáros, L.G. Kinetic analysis of excitation-contraction coupling. J Bioenerg Biomembr 21, 247–266 (1989). https://doi.org/10.1007/BF00812071
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00812071