Summary
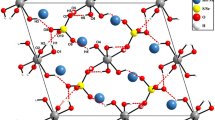
Single crystals of LiFe3+(SeO3)2 were prepared by hydrothermal synthesis. The crystal structure of LiFe3+(SeO3)2 is reported: tetragonal, space group I\(\bar 4\)2d,a=10.649(2) Å,c=9.959(2) Å,V=1129.4 Å3,Z=8, 1268 unique reflections,R=0.037. The structure contains LiFeO8 groups, built up by FeO6 octahedra edgesharing with strongly distorted LiO4 tetrahedra. These LiFeO8 groups share corners with trigonal pyramidal SeO3 groups to form a three dimensional network. The mean bondlengths are 1.994 Å, 2.006 Å and 1.699 Å for Li-O, Fe-O and Se(IV)-O, respectively.
Zusammenfassung
Einkristalle von LiFe3+(SeO3)2 wurden auf hydrothermalem Weg dargestellt und ihre Kristallstruktur bestimmt: tetragonal, Raumgruppe I\(\bar 4\)2d,a=10.649(2) Å,c=9.959(2) Å,V=1129.4 Å3,Z=8, 1268 unabhängige Reflexe,R=0.037. Kennzeichnend für die Atomanordnung sind LiFeO8 Gruppen, die aus FeO6 Oktaedern und mit ihnen kantenverknüpften, stark verzerrten LiO4 Tetraedern aufgebaut sind. Diese LiFeO8 Baueinheiten bilden, mit trigonal pyramidalen SeO3 Gruppen über Ecken verbunden, ein dreidimensionales Netzwerk. Die Mittelwerte der Li-O, Fe-O and Se(IV)-O Abstände sind 1.994 Å, 2.006 Å und 1.699 Å.
Similar content being viewed by others
References
Valkonen J., Koskenlinna M. (1978) Acta Chem. Scand.A32: 603
Hawthorne F. C. (1984) Canad. Min.22: 475
Muilu H., Valkonen J. (1987) Acta Chem. Scand.A41: 183
Giester G., Wildner M. (1991) Mh. Chem.122: 617
Giester G. (1992) Mh. Chem.123: 957
Giester G. (1993) J. Sol. St. Chem.103: 451
Giester G. (1993) Min. Pet.48: 227
Giester G. (1993) Z. Krist.207: 1
Giester G. (1993) Mh. Chem.124: 1107
Giester G. (1994) Mh. Chem. (in press)
Giester G., Pertlik F. (1994) J. Alloys Compounds (in press)
Debaerdemaeker T., Germain G., Main P., Tate C., Woolfson M. M. (1987) MULTAN87. A System of Computer Programs for the Automatic Solution of Crystal Structures from X-ray Diffraction Data, Univs. of Ulm, Germany, Louvain, Belgium, and York, England
Sheldrick G. M. (1976) SHELX-76 program for crystal structure determination. Cambridge, England
Dowty E. (1993) ATOMS 2.3 — a Computer Program for Displaying Atomic Structures. Kingsport, TN
Baur W. H. (1991) Interatomic Distance Predictions for Computer Simulation of Crystal Structures. In: O'Keeffe M., Navrotsky A. (eds.) Structure and Bonding in Crystals, Vol. II, 31. Academic Press, New York
Cocco G., Fanfani L., Zanazzi P. F. (1972) Lithium (3-A). In: Wedepohl K. H. (ed.) Handbook of Geochemistry II-1. Springer, Berlin Heidelberg New York
Hawthorne F. C., Groat L. A., Ercit T. S. (1987) Acta Crystallogr. Sect.C43: 2043
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Giester, G. Crystal structure of LiFe3+(SeO3)2 . Monatsh Chem 125, 535–538 (1994). https://doi.org/10.1007/BF00811845
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00811845