Abstract
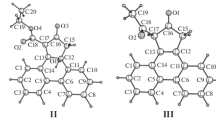
One CO group of the dimethyldihydrophenanthrene mono-Cr(CO)3 complex (1) was photochemically substituted for CS (2) or P(C6H5)3 (3). Separation of all four possible stereoisomers [(R) m (R) b /(S) m (S) b and (R) m (S) b /(S) m (R) b ] of the complexes1–3 was accomplished by two successive chromatographic steps: Separation of the enantiomers on triacetylcellulose was followed by MPLC at low temperatures to yield both epimers (exo andendo). Their configurational assignment is based on optical comparison of the CD-spectra and on1H-NMR-spectroscopy. The kinetics of the biphenyl flip were followed by CD and NMR. The results revealed that the rotational barriers around the biphenyl bond are hardly altered by the substitution of CO for CS or even P(C6H5)3. Whereasexo andendo-isomers of1 and2 are obtained in appr. equal amounts, in the more crowded complex3 theexo-isomer predominates over theendo-form by 80:20%.
Zusammenfassung
Im 4,5-Dimethyl-9,10-dihydrophenanthren-tricarbonylchrom-Komplex (1) wurde eine CO-Gruppe photochemisch gegen CS (2) oder P(C6H5)3 (3) ausgetauscht. Die Trennung aller vier möglichen Stereoisomeren [(R) m (R) b /(S) m (S) b und (R) m (S) b /(S) m (R) b ] der Komplexe1–3 erfolgte in zwei chromatographischen Schritten: Zuerst wurden die Enantiomeren an Triacetylcellulose und anschließend die beiden (exo/endo) Epimeren durch Mitteldruckchromatographie bei tiefen Temperaturen getrennt. Die Konfigurationen konnten auf Grund der CD-Spektren und der1H-NMR-Spektren zugeordnet werden; diese Methoden dienten auch zum Studium der Kinetik der Inversion um die Biphenylbindung (exo/endo-Isomerisierung): Die entsprechenden Rotationsbarrieren werden durch den Austausch von CO gegen CS oder selbst gegen P(C6H5)3 kaum geändert. Dieexo- undendo-Isomeren von1 und2 fallen in gleichen Mengen an, während im sterisch gehinderten Komplex3 dieexo-Form zu 80% überwiegt.
Similar content being viewed by others

References
Part 9:Eyer M, Schlögl K, Widhalm M (1984) Monatsh Chem 115: 1429;
This paper is also part 54 of the series “Stereochemistry of metallocenes” for part 53 see [1 a]
See for instance:Solladie-Cavallo A (1985) Polyhedron 4: 901
Schlögl K (1986) J Organometal Chem 300: 219 and literature cited therein
Kalchhauser H,Schlögl K,Weissensteiner W,Werner A (1983) J Chem Soc Perkin Trans I: 1723;
Schlögl K,Werner A,Widhalm M (1983) J Chem Soc Perkin Trans I: 1731
Tsoy AA, Baranetkaya NK, Setkina VN, Kursanov DN (1981) J Organometal Chem 212: 377
Schöllkopf K, Stezowski J, Effenberger F, (1985) Organometallics 4: 922;
McGlinchey M, Bougeard P, Sayer B, Hofer R, Lock C (1984) J Chem Soc, Chem Comm 1984: 789 and literature cited therein
Schlögl K, Widhalm M (1984) Monatsh Chem 115: 1113
Wittig G, Zimmermann H (1953) Chem Ber 86: 629
Author information
Authors and Affiliations
Additional information
Dedicated to Prof.K. Komarek with cordial wishes on the occasion of the 60th anniversary of his birthday.
Rights and permissions
About this article
Cite this article
Schlögl, K., Werner, A. & Widhalm, M. Biphenyl tricarbonylchromium complexes, part 10. Monatsh Chem 117, 1423–1435 (1986). https://doi.org/10.1007/BF00810752
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00810752