Summary
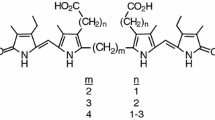
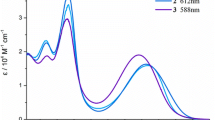
The reduction of mesobiliverdin XIIIα propan-1,3-diyl diester with NaBH4 affords mesobilirubin XIIIα propan-1,3-diyl diester. The same reduction of mesobiliverdin XIIIα methylen diester. The same reduction of mesobiliverdin XIIIα methylen diester affords 8-(2-carboxyethyl)-3,17-diethyl-12-(3-hydroxypropyl)-2,7,13,17-tetramethylbiladien-ac-1,19-(21H, 24H)-dione (MBR-mc). The UV/Vis and1H NMR spectra ofMBR-mc show that its structure in solution is similar to that of the natural bilirubins of the α series.
Zusammenfassung
Reduktion von Mesobiliverdin-XIIIα-propan-1,3-diyl-diester mit NaBH4 liefert Mesobilirubin-XIIIα-propan-1,4-diyl-diester. Die analoge Reaktion des Mesobiliverdin-XIIIα-methylen-diesters führt zu 8-(2-Carboxyäthyl)-3,17-diäthyl-12-(3-hydroxypropyl)-2,7,13,17-tetramethylbiladien-ac-1,19-(21H,24H)-dion (MBR-mc).MBR-mc weist in Lösung (UV/Vis und 1H-NMR Spektren) eine ähnliche Struktur auf wie die der natürlichen Bilirubine der α-Serie.
Similar content being viewed by others
References
Part XVIII of the Series Reactivity of Pyrrole Compounds. Part XVII. Farrera J-A, Anglada MC, Ribó J M (1995) Monatsh Chem126: 355
Falk H (1989) The chemistry of linear oligopyrroles and bile pigments. Springer, Wien New York.
Blanckaert N, Heirwegh K P M, Compernolle F (1976) Biochem J155: 405
Ribó J M, Crusats J, Marco M (1994) Tetrahedron50: 3967
Chem3D Plus™ 3.0, Cambridge Scientific Computing
MCDonagh A F (1979) In: Dolphin D (ed) The porphyrins, vol 6. Academic Press, New York, p 293
Kaplan D, Navon G (1983) Israel J Chem23: 177
Boiadjiev S E, Astine D T, Lightner D A (1994) Tetrahedron: Asymmetry5: 1945
Trull F R, Ma J-S, Landen G L, Lightner D A (1983) Israel J Chem23: 211
Boiadjiev S E, Astine D T, Lightner D A (1995) Tetrahedron Asymmetry6: 901
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Crusats, J., Farrera, J.A. & Ribó, J.M. On the preparation of bilirubins of the natural α series substituted with a propionic acid residue and a hydroxypropyl group. Monatsh Chem 127, 85–89 (1996). https://doi.org/10.1007/BF00807412
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00807412