Summary
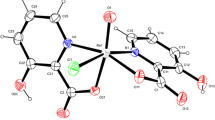
From the reaction systemsCyclam/Mecyclam-KVO3-H2O-HCl-EtOH (Cyclam=1,4,8,11-tetraazacyclotetradecane; Mecyclam=5,5,7,12,12,14-hexamethyl-1,4,8,11-tetraazacyclotetradecane), the first polyvanadates with cations of macrocyclic amines were isolated: (CyclamH2)2-H2V10O28·2H2O, (MecyclamH2)2H2V10O28·6H2O, (CyclamH2)(VO3)2·3H2O, (CyclamH2)(VO3)2·2H2O, and (MecyclamH2)(VO3)2·H2O. The metavanadates with theCyclamH 2+2 cation differ markedly in their IR spectra, obviously due to the different structure of the polyanion. The interruption of dynamic heating at the thermal analysis of the crystallohydrates of dihydrogendecavanadates resulted in the formation of corresponding anhydrous dihydrogendecavanadates.
Zusammenfassung
Aus demCyclam/Mecyclam-KVO3-H2O-HCl-EtOH Reaktionssystem (Cyclam=1,4,8,11-tetraazacyclotetradecane;Mecyclam=5,5,7,12,12,14-hexamethyl-1,4,8,11-tetrazacyclotetradecane) wurden die ersten Polyvanadate mit cyclischen Aminen als Kationen isoliert: (CyclamH2)2V10O28·H2O, (MecyclamH2)2H2V10O28·6H2O, (CyclamH2)(VO3)2·3H2O, (CyclamH2)(VO3)2·2H2O und (MecyclamH2)(VO3)2·H2O. Die zwei Metavanadate mit dem KationCyclamH 2+2 unterscheiden sich wesentlich in ihren Infrarotspektren, was auf unterschiedliche Polyanionstruktur der Metavanadate hinweist. Durch Unterbrechen der dynamischen Erhitzung bei der thermischen Analyse von Kristallhydraten der Dihydrogendekavanadate wurden entsprechende Verbindungen ohne Kristallwasser gewonnen.
Similar content being viewed by others
References
Averbuch-Pouchot MT, Durif A (1994) CR Acad Sci Paris319: 1013
Averbuch-Pouchot MT, Durif A (1994) Eur J Solid State Inorg Chem31: 351
Averbuch-Pouchot MT (1994) Eur J Solid State Inorg Chem31: 557
Day VW, Klemperer WG, Maltrie DJ (1987) J Am Chem Soc109: 2991 and citations therein
Clare BW, Kepert DL, Watts DW (1973) J Chem Soc Dalton: 2481
Román P, José AS, Luque A, Gutierrez-Zorilla JM (1993) Inorg Chem32: 775
Averbuch-Pouchot MT, Durif A (1994) Eur J Solid State Inorg Chem31: 567
Crans DC, Mahroof-Tahir M, Anderson OP, Miller MM (1994) Inorg Chem33: 5586
Suchá V, Sivák M, Schwendt P (1993) Monatsh Chem124: 809
Curtis NF (1964) J Chem Soc 2644
Calcd. for8: C, 17.61; H, 3.99; N, 8.21; V, 37.34%; found: C, 17.80; H, 4.06; N, 8.10; V, 36.95%. Calcd. for9: C, 25.08; H, 5.13; N, 7.31; V, 33.24%; found: C, 24.78; H, 5.16; N, 7.20; V, 32.85%
IR spectra of6: 520m; 563vw; 608m; 625m; 640m; 682w; 762s; 790m; 808m, 852vs; 870m; 890m; 913m; 945s; 975w, and7: 415vw; 440m; 475sh; 504m; 520m; 555m; 595sh; 640m; 685s; 755m; 792s; 855m; 880w; 945s; 975vs; 992s
Zúrková L', Vávra R (1993) Monatsh Chem124: 619
Zabolotskikch AV, Tatyianina IV, Torchenkova EA (1987) Koord Kchim13: 1362
Suchá V, Zúrková L' (1978) Acta Fac RN Univ Comen XXVI: 23
Ulická L' (1976) Chem Zvesti30: 416
Ulická L' (1977) Acta Fac RN Univ Comen Chim XXV: 69
Zúrková L', Ulická L', Suchá V (1978) Proc 7th Conf Coord Chem, Smolenice (Slovakia), p 297
Hawthorne FC, Calvo C (1977) J Solid State Chem22: 157
Palopoli CM, Ethchevery SB, Baran EJ (1989) J Raman Spectr20: 61
Tyrselová J, Kuchta L', Pavelcík F (1995) Acta CrystC51: 1752
Fuchs J, Mahjour S, Pickardt J (1976) Angew Chem Int Ed Engl95: 374
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sucháa, V., Sivák, M. Metavanadates and dihydrogendecavanadates with cations of macrocyclic tetraamines. Monatsh Chem 127, 1–6 (1996). https://doi.org/10.1007/BF00807403
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00807403