Abstract
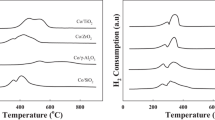
The reduction of cobalt catalysts supported on Al2O3, SiO2, and TiO2 was investigated using a closed system filled with hydrogen gas. Effects of support and metal loading on the rate of reduction were also discussed. The activation energy of reduction increased in the following order: Co/TiO2<Co/Al2O3<Co/SiO2. For different metal loadings, it was found that the catalyst with the higher loading was more readily reducible than that with the lower metal loading. This was confirmed using the results from measurements of particle size, amount of CO adsorbed and activity.
Similar content being viewed by others
References
F.N. Hill and P.W. Selwood, J. Am. Chem. Soc. 71 (1949) 2522.
V.C.F. Holm and A. Clark, J. Catal. 11 (1968) 305.
M. Boudart,Kinetics of Chemical Processes (Prentice-Hall, Englewood Cliffs, 1968).
J.M. Zowtiak and C.H. Bartholomew, J. Catal. 83 (1983) 107.
R.C. Reuel and C.H. Bartholomew, J. Catal. 85 (1984) 63.
B.A. Sexton, A.E. Hughes and T.W. Turney, J. Catal. 97 (1986) 390.
C.R.F. Lund and J.A. Dumesic, J. Phys. Chem. 85 (1982) 3175.
J.W. Niemantsverdriet, A.M. Van Der Kraan and W.N. Delgass, J. Catal. 89 (1984) 138.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Choi, JG. Reduction of supported cobalt catalysts by hydrogen. Catal Lett 35, 291–296 (1995). https://doi.org/10.1007/BF00807185
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00807185