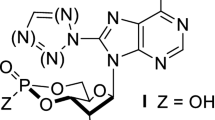
Summary
Various carbocyclic nucleosides withxylo-configuration have been synthesized using ring opening of 5-O-acetyl-1,2-anhydro-3-O-benzylcarba-α-DL-xylo-pentofuranose (6) by thymine, uracil, 4-N-benzoylcytosine, adenine, 6-N-benzoyladenine, and 2-amino-6-chloropurine in alkaline medium. For this purpose, the use of triethylaluminum is introduced into carbanucleoside chemistry. The new method proved to be superior over the application of sodium hydride and potassium or caesium carbonate.
Zusammenfassung
Ringöffnung von 5-O-Acetyl-1,2-anhydro-3-O-benzylcarba-α-DL-xylo-pentofuranose (6) mit Thymin, Uracil, 4-N-Benzoylcytosin, Adenin, 6-N-Benzoyladenin und 2-Amino-6-chlorpurin in alkalischem Medium ergab die entsprechendenxylo-konfigurierten carbocyclischen Nucleoside. Als Hilfsbase wurde Triethylaluminium verwendet, was deutliche Vorteile gegenüber der Verwendung von Natriumhydrid und Kalium- oder Cäsiumcarbonat bezüglich Produktreinheit und Ausbeute bietet.
Similar content being viewed by others
References
For an overview see: a) Marquez VE, Lim M-I (1986) Med Res Rev6: 1;
Borthwick AD, Biggadike K (1992) Tetrahedron48: 571;
Agrofoglio L, Suhas E, Farese A, Condom R, Challand SR, Earl RA, Guedj R (1994) Tetrahedron50 10611
Isono K (1988) J Antibiotics41: 1711;
Vince R, Hua M, Brownell J, Daluge S, Lee F, Shannon WM, Lavelle GC, Qualls J, Weislow OS, Kiser R, Canonico PG, Schultz RH, Narayanan VL, Mayo JG, Shoemaker RH, Boyd MR (1988) Biochem Biophys Res Commun156: 1046;
White EL, Parker WB, Macy LJ, Shaddix SC, McCaleb G, Secrist III JA, Vince R, Shannon WM (1989) Biochem Biophys Res Commun161: 393;
Vince R, Hua M (1990) J Med Chem33: 17;
Taylor SJC, Sutherland AG, Lee C, Wisdom R, Thomas S, Roberts SM, Evans C (1990) J Chem Soc Chem Commun 1120;
Evans C, McCague R, Roberts SM, Sutherland AG (1991) J Chem Soc Perkin Trans 1, 656;
Robins RK, Revankar GR (1988) In: DeClercq E, Walker RT (eds) Antiviral Drug Development. Plenum, New York, p 11
Shaw G, Warrener RN (1958) J Chem Soc: 153
Tseng CKH, Marquez VE (1985) Tetrahedron Lett26: 3669;
Biggadike K, Borthwick AD, Evans D, Exall AM, Kirk BE, Roberts SM, Stephenson L, Youds P (1988) J Chem Soc Perkin Trans 1, 549
Hutchison A, Grim M, Chen J (1989) J Heterocyclic Chem26: 451;
Yoshikawa M, Nakae T, Cha BC, Yokokawa Y, Kitagawa I (1989) Chem Pharm Bull37: 545;
Yoshikawa M, Okaichi Y, Cha BC, Kitagawa I (1990) Tetrahedron46: 7459;
reference 1 c and references 35–45 cited therein
Baumgartner H, Marschner C, Pucher R, Griengl H (1991) Tetrahedron Lett32: 611
Eichberger G, Penn G, Faber K, Griengl H (1986) Tetrahedron Lett27: 2843
Meinwald J, Seidel MC, Cadoff BC (1958) J Am Chem Soc80: 6303
Baumgarten RJ (1966) J Chem Education43: 398
Madhavan GVB, McGee DPC, Rydzewski RM, Boehme R, Martin JC, Prisbe EJ (1988) J Med Chem31: 1798
Biggadike K, Borthwick AD, Exall AM, Kirk BE, Roberts SM, Youds P (1987) J Chem Soc Chem Comm 1083
Overman LE, Sugai S (1985) J Org Chem50: 4154
For detailed information see: Balzarini J, Baumgartner H, Bodenteich M, De Clercq E, Griengl H (1989) Nucleosides & Nucleotides8: 855
Vince R, Brownell J, Daluge S (1984) J Med Chem27: 1358
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kapeller, H., Baumgartner, H., Marschner, C. et al. The ring opening of cyclopentene oxides by pyrimidines and purines as a pathway to carbocyclic nucleoside analogues. Monatsh Chem 128, 953–960 (1997). https://doi.org/10.1007/BF00807105
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00807105