Summary
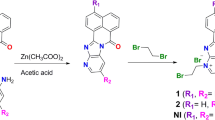
This work is an extension of our previous work (Hall et al., 1993) on the synthesis and cytotoxic activity of boronated peptides. The aim of this work was to carry out structural modifications of the amine terminal in compounds1 and2, to increase water solubility, and its effect on the cytotoxicity to tumor cell lines. Surprisingly, only compounds4,7 and8 were more water soluble than the parent compounds. With the exception of compound4, the new derivatives were generally less effective than the parent compounds (1 and2). There was no apparent correlation between structure and activity. Cytotoxic effect was more pronounced in single cell suspended cells. The growth of solid tumor cell lines was not significantly reduced. The most active derivative, (methanamine)dihydro[[[1-(phenylmethyl)-2-methylamino-2-oxoethyl]amino]carboxy]boron (4), inhibited DNA, RNA, and protein synthesis in Tmolt3 cells. Enzymatic activities, e.g., DNA polymeraseα, m-RNA polymerase, PRPP amidotransferase, carbamyl phosphate synthetase, TMP-kinase, TDP-kinase, dihydrofolate reductase, and ribonucleoside reductase were reduced after 60 min incubation with4. d(TTP) and d(CTP) pool levels were also reduced by 60 min incubation with4.
Similar content being viewed by others
References
Anderson KM, Mendelson IS, Guzik G (1975) Solubilized DNA-dependent nuclear RNA polymerases from the mammary glands of late-pregnant rats. Biochem Biophys Acta 383: 56–66
Appel SH (1968) Purification and kinetic properties of brain orotidine 5′-phosphate decarboxylase. J Biol Chem 243: 3924–3929
Archibald RM (1944) Determination of citrulline and allontoin and demonstration of citrulline in blood plasma. J Biol Chem 156: 121–142
Bagnara AS, Finch LR (1972) Quantitative extraction and estimation of intracellular nucleoside triphosphates inEscherichia coli. Anal Biochem 45: 24–34
Becker JH, Lohr GW (1979) Inosine 5′-phosphate dehydrogenase activity in normal and leukemic blood cells. Klin Wochenschr 57: 1109–1115
Cadman E, Heimer R, Benz C (1981) The influence of methotrexate pretreatment on 5-fluorouracil metabolism in L1210 cells. J Biol Chem 256: 1695–1704
Christopherson RI, Yu ML, Jones ME (1981) An overall radioassay for the first three reactions ofde novo pyrimidine synthesis. Anal Biochem 11: 240–249
Das MK, Roy S (1985) Convenient and high-yield synthesis of triphenylphosphine-cyanoborane and dihaloboranes. Synth React Inorg Met-Org Chem 15: 53–59
Eichler DC, Fisher PA, Korn D (1977) Effect of calcium on the recovery distribution of DNA polymeraseα from cultured human cells. J Biol Chem 252: 4011–4014
Geran RJ, Greenburg NH, MacDonald MM, Schumacher AM, Abbott BJ (1972) Protocols for screening chemical agents and natural products against animal tumors and other biological systems. Cancer Chemother Rep 3: 9–24
Hall IH, Carlson GL, Abernathy GS, Piantadosi C (1974) Cycloalkanones: antifertility activity. J Med Chem 17: 1253–1257
Hall IH, Hall ES, Miller MC, Sood A, Spielvogel BF (1993) The effects of boron containing peptides on L1210 lymphoid leukemia metabolism. Amino Acids 4: 287–302
Ho YK, Hakala T, Zakrzewski SF (1972) 5-(l-adamantyl) pyrimidines as inhibitors of folate metabolism. Cancer Res 32: 1023–1028
Hunting D, Henderson JF (1982) Determination of deoxyribonucleoside triphosphates using DNA polymeraseα: a critical evaluation. Can J Biochem 59: 723–727
Kalman SM, Duffield PH, Brzozwski TJ (1966) Purification and properties of a bacterial carbamyl phosphate synthetase. J Biol Chem 241: 1871–1877
Kampf A, Barfknecht RL, Schaffer PJ, Osaki S, Mertes MP (1976) Synthetic inhibitors ofEscherichia coli calf thymus and Ehrlich ascites tumor thymidylate synthetase. J Med Chem 19: 903–908
Koritz SB, Gohen PP (1954) Colorimetic determination of carbamyl amino acid and related compounds. J Biol Chem 209: 145–150
Koster H (1983) Methoden der Organischen Chemie (Houben-Weyl), Band 13/36. Organobor Verbindungen II. G Thieme, Stuttgart, pp 424–680
Leibovitz AL, Stinson JC, McComb III WB, McCoy CE, Mazur KC, Mabry ND (1976) Classification of human colorectal adenocarcinoma cell lines. Cancer Res 36: 4562–4569
Liao L, Kupchan SM, Horwitz SB (1976) Mode of action of the antitumor compound bruceantin, an inhibitor of protein synthesis. Mol Pharmacol 12: 167–176
Lowry OH, Rosebrough J, Farr AL, Randall RJ (1951) Protein measurement with Folin phenol reagent. J Biol Chem 193: 265–275
Maley F, Ochoa S (1958) Enzymatic phosphorylation of deoxycytidylic acid. J Biol Chem 233: 1538–1543
Mamaril FP, Dobrjasky A, Green S (1970) A rapid method for isolation of nuclei of Ehrlich ascites tumor cells. Cancer Res 30: 352–356
Moore EC, Hurlbert RB (1966) Regulation of mammalian deoxyribonucleotide biosynthesis of nucleotides or activators and inhibitors. J Biol Chem 241: 4802–4809
Pera Jr MF, Rawlings CJ, Shackleton J, Roberts JJ (1981) Quantitative aspects of the formation and loss of DNA interstrand crosslinks in Chinese hamster cells following treatment with cis-diamminodichloroplatinum (II) cis-platin. Biochem Biophys Acta 655: 152–166
Spielvogel BF, Sood A, Tomasz J, Shaw BR, Burnham B, Hall IH (1992) One-step synthesis of boronated purines, nuclosides and nucleotides and their pharmacological properties. Boron USA III, Pullman, Washington, Abstract 54
Spielvogel BF, Sood A, Tomasz J, Shaw BR, Karthikeyan S, Powell W, Laster B, Brugger RM, Coderre J (1993) Boronated peptides and nucleic acid components for NCT. In: Soloway AH, Barth RF, Carpenter DE (eds) Advances in neutron capture therapy. Plenum Press, New York, pp 361–365
Zhao Y, Hall IH, Oswald CB (1987) Anti-malarial agents III. Mechanism of action of artersurate againstPlasmodium berghi infection. Chem Pharm Bull 35: 2052–2061
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Karthikeyan, S., Sood, A., Tomasz, J. et al. Cytotoxicity of boron containing dipeptide analogs. Amino Acids 8, 323–335 (1995). https://doi.org/10.1007/BF00806550
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00806550