Abstract
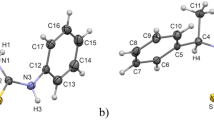
Reaction of 7-azaindole with activated malonates (1) results in the formation of the tricyclic mesoionic diaza-acenaphthylene compounds2. Hydrogenation of2a with platin as catalyst leads to the saturated betaine4. The structure assignment was established by1H-13C-NMR shift correlation and decoupling experiments.
Similar content being viewed by others
Literatur
XV. Mitt.:Kappe Th., Ravai Y. Stadlbauer W., Monatsh. Chem.114, 227 (1983).
Review:Friedrichsen G., Böttcher A., Kappe Th., Heterocycles19, 1083 (1982).
Kappe Th., Lube W., Monatsh. Chem.102, 781 (1971).
Galvez C., Viladoms P., J. Heterocycl. Chem.19, 665 (1982).
Thonhofer K., Dissertation, Univ. Graz, 1978;Wildpanner H., Dissertation Univ. Graz 1982.
Kappe Th., Lube W., Chem. Ber.112, 3424 (1979).
Ziegler E., Junek H., Monatsh. Chem.90, 762 (1959):Ziegler E., Gelfert K., ibid.90, 858 (1959);Ziegler E., Nölken E., Gelfert K., Salvador R., ibid.92, 814 (1961).
Sterk H., Suschnigg J. J., Thonhofer K., Z. Naturforsch.31a, 793 (1976).
Author information
Authors and Affiliations
Additional information
Herrn Prof. Dr.E. Schauenstein, Vorstand des Institutes für Biochemie der Universität Graz, mit den besten Wünschen zum 65. Geburtstag gewidmet.
Rights and permissions
About this article
Cite this article
Stadlbauer, W., Ravai, Y., Sterk, H. et al. Mesoionische Sechsringheterocyclen, XVI Syntheses von 2 a, 5 a-Diaza-acenaphthylen Mesoionen. Monatsh Chem 114, 965–972 (1983). https://doi.org/10.1007/BF00799956
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00799956