Abstract
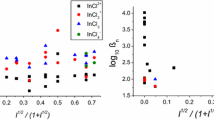
Potentiometric and conductometric studies on the zinc, cadmium, lead—methylene bis thio acetic acid system in aqueous media reveal the formation of 1:1 and 1:2 (M:L) complexes in thepH range 3.4–5.1. Their stability constants have been determined at 20°, 30° and 40 °C by applyingCalvin andMelchior's extension ofBjerrum's method. The overall changes in thermodynamic function ΔH, ΔG and ΔS accompanying complexation have been evaluated at 30 °C.
Zusammenfassung
Potentiometrische und konduktometrische Untersuchungen an Zink-, Cadmium- und Blei-Methylen-bis-thioessigsäure-Systemen in wäßrigem Medium zeigten die Bildung von 1:1 und 1:2 (M:L)-Komplexen impH-Bereich 3,4–5,1. Die Stabilitätskonstanten wurden bei 20°, 30° und 40 °C mittels derCalvin-Melchior-Methode (erweiterteBjerrum-Methode) bestimmt. Die thermodynamischen Parameter ΔH, ΔG und ΔS der Komplexierung wurden für 30 °C ermittelt.
Similar content being viewed by others
References
Saxena R. S., Gupta K. C., Z. phys. Chem.241, 169 (1969).
Saxena R. S., Pratap Singh, Monatsh. Chem.101, 512 (1970).
Saxena R. S., Chaturvedi U. S., J. Inorg. Nuclear Chem.33, 3597 (1971).
Saxena R. S., Gupta K. C., Z. Naturforsch.24 B, 795 (1969).
Calvin M., Melchior N. C., J. Amer. Chem. Soc.70, 3270 (1948).
Bjerrum J., Metal Amine Formation in Aqueous Solutions. Copenhagen: P. Hasse & Sons. 1941.
Irving H.,Rossotti H. S., J. Chem. Soc.1953, 3397.
Schroder K. N., Acta Chem. Scand.20, 1401 (1966).
Yatsimirskii K. B., Vasilev V. P., Instability Constants of Complex Compound. Oxford: Pergamon Press. 1960.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Saxena, R.S., Parikh, R.D. & Gupta, K.C. Electrochemical studies on the composition, stabilities and thermodynamics of Zn+2, Cd+2 and Pb+2 complexes with methylene bis thio acetic acid. Monatsh Chem 115, 141–146 (1984). https://doi.org/10.1007/BF00798404
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00798404