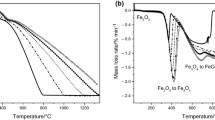
Conclusions
The specific rate of additional reduction of the oxides films on the particles of iron in a falling layer of the powder in the temperature range 800–1000°C calculated for the initial specific surface was 0.036–0.078 g[O]/m2· sec. The activation of the reduction process was equal to 54.16 kJ/mole.
The rate of additional reduction of the iron powder in the falling layer is high. For example, in the temperature range 800–1000°C it is 1.2–3.1% [O]/sec.
The experimental results as well as the comparison with the available data make it possible to conclude that the process of reduction of the surface oxides in the falling layer in the temperature range 800–1000°C takes place in the kinetic range of reaction and is limited by the reaction of interaction of hydrogen with the surface oxides of iron.
Similar content being viewed by others
Literature cited
V. B. Akimenko, V. Ya. Bulanov, and V. V. Rukin, Iron Powders [in Russian], Nauka, Moscow (1982).
V. A. Maslov, D. A. Kapustin, Yu. A. Aleksandrov, and A. M. Kulakov, “A new principal of annealing metallic powders and its design features,” in: New Methods of Producing Metallic Powders [in Russian] Institute of Problems of Materials Science, Academy of Sciences of the Ukrainian SSR, Kiev (1981), pp. 27–31.
V. A. Maslov, Yu. A. Aleksandrov, and D. A. Kapustin, “Detailed examination of reduction of annealing of iron powders in a falling layer,” in: New Methods of Producing Metallic Powders [in Russian], Institute of Problems of Materials Science, Academy of Sciences of the Ukrainian SSR, Kiev (1985), pp. 72–75.
V. A. Maslov, “A new principle of reduction of annealing of metallic powders,” in: Proceedings of 7th International Powder Metallurgy Conference, Ostrava, Czechoslovakia (1987), pp. 21–28.
V. A. Maslov, Yu. A. Aleksandrov, and D. A. Kapustin, “Experimental examination of the dynamics of movement and heating of dispersed material in a falling layer,” in: Heat and Mass Transfer-7: Proceedings of 7th All-Union Conference on Heat and Mass Exchange [in Russian], Vol. 5, Part 1, Minsk (1984), pp. 117–121.
Z. R. Gorbis, Heat Exchange and Hydrodynamics of Dispersed Continuous Flows [in Russian], Énergiya, Moscow (1977).
A. A. Shraiber, V. N. Minotin, and V. P. Yatsenko, Hydrodynamics of Two Component Flows with a Solid Polydispersed Material [in Russian], Naukova Dumka, Kiev (1980).
O. Levenspiele, Engineering Formulation of Chemical Processes [Russian Translation], Khimiya, Moscow (1969).
V. V. Kafarov, Cybernetics Methods in Chemical Technology [in Russian], Khimiya, Moscow (1971).
L. S. Bogdandi and G. Yu. Engel, Reduction of Iron Ore [Russian Translation], Metallurgiya, Moscow (1970).
Author information
Authors and Affiliations
Additional information
Translated from Poroshkovaya Metallurgiya, No. 9-(345), pp. 1–5, September, 1991.
Rights and permissions
About this article
Cite this article
Maslov, V.A., Aleksandrov, Y.A. Mechanism of additional reduction of metallic powders in a falling layer. Powder Metall Met Ceram 30, 713–716 (1991). https://doi.org/10.1007/BF00794205
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00794205