Conclusions
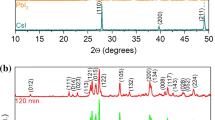
The reaction of cubic boron nitride with the gaseous phase of titanium iodides (process duration 0.25–2.0 h, total pressure of titanium iodides 100 Pa) results in the preferential formation of TIN in the temperature range 900–1200°K and of titanium nitride and titanium diboride at 1200–1300°K.
Similar content being viewed by others
Literature cited
V. P. Bondarenko and A. E. Shilo, “Thermodynamic investigation of the reactions of boron nitride with transition metals,” Sintetich. Almazy, No. 2, 13–18 (1977).
V. P. Bondarenko, A. P. Khalepa, and E. S. Cherepenina, “Investigation of the reactions of cubic boron nitride with transition metals and their carbides,” Sintetich. Almazy, No. 4, 22–25 (1978).
Yu. V. Naidich, A. S. Vishnevskii, G. A. Kolesnichenko, et al., “Metallization of cubic boron nitride,” in: Wetting and Surface Properties of Melts and Solids [in Russian], Naukova Dumka, Kiev (1967), pp. 179–181.
I. I. Gavrilov, A. I. Evstyukhin, V. A. Shulov, and M. M. Kozlov, “Thermodynamics and kinetics of the process of chemical transport of tantalum in the tantalum-iodine system,” Zh. Fiz. Khim.,57, No. 6, 1347–1350 (1983).
R. F. Rolsten, Iodide Grade Metals and Metallic Iodides [in Russian], Metallurgiya, Moscow (1968).
M. Kh. Karapetyants and M. L. Karapetyants, Principal Thermodynamic Constants of Inorganic and Organic Substances [in Russian], Khimiya, Moscow (1968).
A. V. Kurdyumov and A. N. Pilyankevich, Phase Transformations in Carbon and Boron Nitride [in Russian], Naukova Dumka, Kiev (1979).
A. A. Rusakov, X-Ray Diffraction Investigations of Metals [in Russian], Atomizdat, Moscow (1977).
Author information
Authors and Affiliations
Additional information
Translated from Poroshkovaya Metallurgiya, No. 10(262), pp. 57–59, October, 1984.
Rights and permissions
About this article
Cite this article
Vesna, V.T., Maslov, V.P. Reaction of cubic boron nitride with titanium iodides. Powder Metall Met Ceram 23, 787–789 (1984). https://doi.org/10.1007/BF00792146
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00792146