Abstract
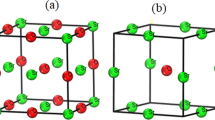
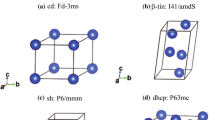
The results of a low-temperature spectroscopic investigation of crystals of the highest hydrate of perchloric acid have been presented. A brief review of the literature data on the problem of the structure of crystals of HClO4·5.5H2O (I) has been given. It follows from the review that the negative results obtained by Mootz et al. [J. Am. Chem. Soc., Vol. 109, No. 4, 1200 (1987)] when they refined a clathrate test model of the cubic structure of I is attributable to the mesomorphic structure of the crystals, which is characteristic of a plastic phase. The results of an analysis of the IR and Raman spectra of the optically anisotropic α plastic phase of I and the isotropic β plastic phase have made it possible to specify some details of the clathrate model. The vibrational spectra of deuterated and undeuterated solid solutions of I with components of the acid-water system have been interpreted in the framework of the improved clathrate model. Laws governing the structure of the labile polyhedrons in the water-cation host structure and the formation of the anion sublattice, which is insensitive to variation of the composition, have been revealed for the two phases of variable composition α-HClO4·nH2O (n=5.4 to 5.6 at −180°C) and β-HClO4·nH2O (n=5.2 to 6.0 at −180°C).
Similar content being viewed by others
Literature Cited
A. I. Karelin, Izv. Akad. Nauk SSSR, Ser. Khim., No. 1, 12–17 (1988).
D. E. Mootz, E.-J. Oellers, and M. Wiebcke, J. Am. Chem. Soc.,109, No. 4, 1200–1202 (1987).
H. J. Van Wyk, Z. Anorg. Chem.,48, No. 1, 1–52 (1906).
G. Mascherpa, Rev. Chim. Miner.,2, 379–433 (1965).
U. Frese, T. Iwasita, W. Schmickler, and U. Stimming, J. Phys. Chem.,89, No. 7, 1059–1064 (1985).
T. H. Huang, R. A. Davis, U. Frese, and U. Stimming, J. Phys. Chem.,92, No. 24, 6874–6876 (1988).
J. N. Sherwood (editor), The Plastically Crystalline State: Orientationally Disordered Crystals, Wiley, New York (1979).
D. Gratias, Usp. Fiz. Nauk,156, No. 2, 347–364 (1988).
G. M. Zaslavskii, R. Z. Sagdeev, D. A. Usikov, and A. A. Chernikov,Ibid., No. 2, 193–251 (1988).
V. Ya. Rosolovskii, Chemistry of Anhydrous Perchloric Acid [in Russian], Nauka, Moscow (1966), p. 53.
A. S. Pavia and P. A. Giguere, J. Chem. Phys.,52, No. 7, 3551–3554 (1970).
E. Chemouni, M. Fournier, J. Roziere, and J. Potier, J. Chim. Phys.-Chim. Biol.,67, No. 3, 517–521 (1970).
G. A. Geffrey, in: J. L. Davies (editor), Inclusion Compounds, Vol. 1, Academic Press, London (1984), pp. 135–190.
J.-O. Lundgren and I. Olovsson, in: P. Schuster et al., (editors), The Hydrogen Bond, Vol. 1, North-Holland, Amsterdam (1976), pp. 471–527 (1976).
J. Almlöf, Chem. Scr.,3, No. 2, 73–79 (1973).
C. I. Ratcliffe and D. E. Irish, Can. J. Chem.,62, No. 6, 1134–1144 (1984).
H. A. Hardin and K. B. Harvey, Spectrochim. Acta,29A, No. 6, 1139–1151 (1973).
E. Whalley, in: P. Schuster et al., (editors), The Hydrogen Bond, Vol. 3, North-Holland, Amsterdam (1976), pp. 1425–1466.
A. I. Karelin, Z. I. Grigorovich, and V. Ya. Rosolovskii, Spectrochim. Acta,31A, No. 5/6, 765–775 (1975).
G. P. Johari and H. A. M. Chew, Philos. Mag.,49, No. 3, 281–294 (1984).
G. P. Johari and H. A. M. Chew,Ibid., No. 6, 647–660 (1984).
Additional information
Institute of New Chemical Problems, Academy of Sciences of the USSR. Translated from Zhurnal Strukturnoi Khimii, Vol. 32, No. 2, pp. 51–61, March–April, 1991.
Rights and permissions
About this article
Cite this article
Karelin, A.I. Solid solutions, clathrate structures, and vibrational spectra of plastic crystals of HClO4·5.5H2O. J Struct Chem 32, 199–208 (1991). https://doi.org/10.1007/BF00777188
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00777188