Summary
-
1.
A study was made of the oxidation of boron carbide and carbon black by different oxidizers: mixtures of sulfuric acid with potassium bichromate, sulfuric acid with potassium permanganate, sulfuric, nitric, and perchloric acids with potassium bichromate, etc. It is shown that the best oxidizing mixture for the removal of free carbon from boron carbide is one containing sulfuric, nitric, and perchloric acids with potassium bichromate.
-
2.
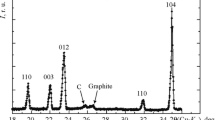
A study was made of the oxidation of boron carbide powders with different amounts of total carbon and boron by oxygen at different temperatures. It is shown that the oxidation of boron carbide (and free carbon) begins at 700°C. The formation of a B2O3 oxide film prevents the full oxidation of boron carbide at 1100°C. The complete oxidation of boron carbide by oxygen is only observed at 1200–1300°C. In the case of two boron carbide samples subjected to oxidation by oxygen, the higher oxidation intensity will be shown by the sample with the smaller value of the coefficient k=Bcomb./Ctotal.
Similar content being viewed by others
Literature cited
T. N. Nazarchuk, Zh. Neorgan. Khim.,4, 2665 (1959).
Yu. A. Klyachko and M. M. Shapiro, Zavodsk. Lab.,14, 2549 (1948).
S. M. Gutman, Zavodsk. Lab.,13, 1403 (1947).
N. M. Popova and L. V. Zaslavskaya, Zavodsk. Lab.,21, 1285 (1955).
A. M. Rekov and G. V. Samsonov, Poroshkovaya Met., 5, 82 (1961).
F. Smith, Analyt. chim. acta. 13, 15 (1955).
H. Moissan, C. r., 118, 556 (1894).
R. Ridgway, Trans. Electrochem. Soc., 65, 117 (1934).
G. A. Meerson and G. V. Samsonov, Zavodsk. Lab.,12, 1423 (1950).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nazarchuk, T.N., Mekhanoshina, L.N. The oxidation of boron carbide. Powder Metall Met Ceram 3, 123–126 (1964). https://doi.org/10.1007/BF00774482
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00774482