Abstract
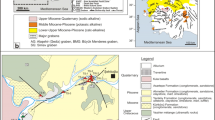
Chaves thermal waters (76°C) are the most important external manifestations of low-temperature geothermal systems occurring in Portuguese mainland. They are related to crystalline granitic rocks. This paper describes the use of the environmental isotopic composition (oxygen-18, deuterium, and tritium) of hot and cold waters of the Chaves area as an important hydrogeological tool to solve specific problems arising in the appraisal of the geothermal resources of the area (e.g., origin and “age” of waters, recharge area, and underground flow paths).18O and D analyses seem to confirm the meteoric origin of Chaves thermal waters. The local altitude dependence of meteoric waters was determined by18O and D analyses of superficial and shallow groundwaters of the Chaves plain and its bordering mountains. Chaves thermal waters seem to be related to meteoric waters, infiltrated on the highest topography areas (Bolideira granitic outcrop NE Chaves), that percolate at great depth and emerge in a discharge area at lower altitude (Chaves plain). Chaves thermal waters showing little oxygen-18 shift and low tritium concentrations could be considered external manifestations of an old geothermal system in which the isotopic water-rock interaction is adjusted to equilibrium.
Similar content being viewed by others
References
Afonso AR, Santos FM, Dupis A, Rio MA, and Victor LM (1993) Geophysical methods in the evaluation of geothermal resources. Application to Chaves (Portugal) geothermal field. Minutes of the meeting 24–29 October, 1993, S. Pedro do Sul Spa, Portugal. IAH-Commission on Mineral and Thermal Waters/Internal Communications 1993. Edited by B Zuurdeeg. Vianen, The Netherlands. Enclosure 10. 13 pp
Aires-Barros L, Graca RC, and Marques JM (1991) Nota preliminar sobre a geoquimica das aguas termais de Chaves. Geocienc Rev Univ Aveiro 6:59–69
Carvalho MR and Almeida C (1989) HIDSPEC, um programa de especiação e cálculo de equilibrios água/rocha. Geocienc Rev Univ Aveiro 4:1–22
Craig H (1961) Isotopic variations in meteoric waters. Science 133:1702–1703
Criaud A and Fouillac C (1986) Etude des eaux thermominérales carbogazeuses du Massif Central Français. II. Comportement de quelques métaux en trace, de l'arsenic, de l'antimoine et du germanium. Geochim Cosmochim Acta 50:1573–1582
Fournier RO (1977) Chemical geothermometers and mixing models for geothermal systems. Geothermics 5:41–50
Fournier RO and Potter RW II (1979) Magnesium correction to the Na-K-Ca chemical geothermometer. Geochim Cosmochim Acta 41:1543–1550
IAEA (International Atomic Energy Agency, Vienna) (1981) Stable isotope hydrology. Deuterium and oxygen-18 in the water cycle. Technical Reports Series No. 210. IAEA, Vienna. 340 pp
Maisonnneuve J and Risler JJ (1979) La ceinture péri-alpine “carbogazeuse” de l'Europe occidentale. Bull Bur Rech Geol Miniers Sect III. 2:109–120
Michard G (1990) Behaviour of major elements and some trace elements (Li, Rb, Cs, Sr, Fe, Mn, W, F) in deep hot waters from granitic areas. Chem Geol 89:117–137
Truesdell AH (1975) Summary of Section III. Geochemical techniques in exploration. Proceedings, second United Nations symposium on the development and use of geothermal resources, San Francisco, California. Edited by Lawrence Berkeley Laboratory, University of California, pp 53–79
UTAD (Universidade de Trás-os-Montes e Alto Douro, Portugal) (1992) Report on the hydrogeology of the geothermal pole of Chaves. Final report for JOULE I programme. UTAD, Department of Geology, Vila Real, Portugal. 40 pp
White DE (1970) Geochemistry applied to the discovery, evaluation, and exploitation of geothermal energy resources. Proceedings, First United Nations Symposium on the Development and Use of Geothermal Resources, Pisa, Italy. Geothermics Special Issue 2, Part 1. pp 58–80
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Aires-Barros, L., Marques, J.M. & Graça, R.C. Elemental and isotopic geochemistry in the hydrothermal area of Chaves, Vila Pouca de Aguiar (northern Portugal). Geo 25, 232–238 (1995). https://doi.org/10.1007/BF00766751
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00766751