Abstract
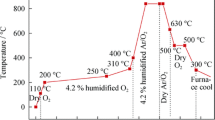
The measurements of MS-TPD, TPRS, the electrical conductivity and kinetics on Ba2YCu3O7-8 show that the catalytic activity of CO oxidation is closely related to properties such as the amounts and sites of oxygen, and electrical conductivity. Based on the experimental results a reaction mechanism has been suggested.
Similar content being viewed by others
References
C.W. Chu et al., Phys. Rev. Lett. 58 (1987) 405.
R.T. Cava, B. Batlogg, C.H. Chen, E.A. Rietman, S.M. Zahurak and D. Werder, Nature 329 (1987) 423:
W.I.F. David et al., Nature 327 (1987) 310.
Q.W. Zhou, A.R. Jiang et al., Acta Physico-chimica Sinica 4 (1988) 229.
W.J. Moore, Physical Chemistry (second edition) (Prentice-Hall Inc. Englewood Cliffs) 549.
T. Nakamura, M. Misono and Y. Yoneda, Bull. Chem. Soc. Jpn. 55 (1982) 394.
J. Haber, Z. Chem. 13 (1973) 241.
P. Bordet et al., Nature 327 (1987) 687.
T.K. Worthington, W.J. Gallagher and T.K. Dinger, Phys. Rev. Lett. 59 (1987) 1160.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
AnRen, J., Ying, P., QinWei, Z. et al. The catalytic oxidation of CO on superconductor Ba2YCu3O7–8 . Catal Lett 3, 235–245 (1989). https://doi.org/10.1007/BF00766399
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00766399