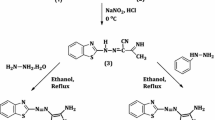
Abstract
A new heterocyclic base- 2-methylfuro[2,3-g]benzothiazole — and cyanine dyes with residues of this base were synthesized. It is shown that introduction of a furo[2,3] group in the 6,7 positions of the benzothiazole residue in the cyanines leads to a smaller bathochromic effect than the introduction of a thieno[2,3] and, particularly, a benzo group in the same positions.
Similar content being viewed by others
Literature cited
Z. I. Moskalenko and M. A. Al'perovich, Khim. Geterotslkl. Soedin., 626 (1967).
M. Descamps and F. Henaux, French Patent No. 1537206 (1968); Chem. Abstr.,71(13), 430 (1969).
L. M. Sharkova, N. F. Kucherova, and V. A. Zagorevskii, Khim. Geterotsikl. Soedin., 1075 (1972).
L. G. Brooker and F. L. White, J. Amer. Chem. Soc.,57, 480 (1935).
F. M. Hamer, J. Chem. Soc., 3160 (1928).
F. M. Hamer, J. Chem. Soc., 2598 (1929).
O. Bloch and F. M. Hamer, Phot. J.,70, 374 (1930).
F. M. Hamer, J. Chem. Soc., 1480 (1956).
P. I. Abramenko and V. G. Zhiryakov, Khim. Geterotsikl. Soedin., 1611 (1972).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 12, pp. 1603–1606, December, 1975.
Rights and permissions
About this article
Cite this article
Abramenko, P.I., Zhiryakov, V.G. & Ponomareva, T.K. Polymethine dyes — Furo[2,3-g]benzothiazole derivatives. Chem Heterocycl Compd 11, 1361–1364 (1975). https://doi.org/10.1007/BF00764525
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00764525