Conclusion
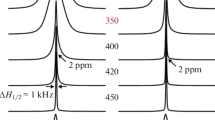
All the oxide fluorides studied show intensive diffusion of the fluoride ions, and this is responsible for their high electrical conductivity. The anisotropy of the magnetic shielding of the fluorine nuclei in the “rigid” lattice has been determined from the field dependence of the second moment. In the high-temperature range, the anisotropy was measured from the form of the resonance line. The coincidence of the values of the anisotropy at high and low temperatures shows that in stoichiometric lanthanum oxide fluoride below the temperature of the phase transformation into the cubic modification, the fluoride ions move only through the equivalent positions of the fluorine sublattice. Analysis of the chemical shifts and their anisotropy shows that the movement of the fluoride ions has vacancy character. Above Tc, the exchange of positions with oxygen is possible. The behavior of the spectra in the intermediate range indicates high dynamic nonuniformity of the fluoride ions, brought about by the existence of regions with an increased fluorine mobility, which apparently have a structure similar to that of the nonstoichiometric lanthanum oxide fluorides with an increased fluorine concentration. The replacement of oxygen by fluorine at first leads to a sharp increase in the mobility of the fluoride ions (x=0.04), after which it has practically no influence up to x=0.143. It is not excluded that the increase in the mobility is related not to the excess of fluorine but to a rearrangement of the crystal structure. Judging from the behavior of the spectral lines in the intermediate range, nonstoichiometric lanthanum oxide fluorides are dynamically more uniform.
Similar content being viewed by others
Literature cited
P. McGeehin and A. Hooper, J. Mater. Sci.,12, 1 (1977).
A. Pelloux, P. Fabry, and C. C. Deportes, Compt. Rend. Acad. Sci.,276c 241 (1973).
V. A. Levitskii, G. M. Balak, M. L. Kovba, and P. B. Narchuk, Summaries of Papers Presented at the Third Urals Conference on High-Temperature Physical Chemistry and Electrochemistry [in Russian], Sverdlovsk (1981), p. 265.
M. Kleitz, in: Fast Ion Transport in Solids, W. Van Gool, ed., Plenum Press, New York (1973), p. 602.
W. Klemm and H. A. Klein, Z. Anorg. Allgem. Chem.,248, 167 (1941).
W. H. Zachariasen, Acta Crystallogr.,4, 231 (1951).
K. Niihara and S. Yajima, Bull. Chem. Soc. Jpn.,44, 143 (1971).
K. Niihara and S. Yajima, Bull. Chem. Soc. Jpn.,45, 20 (1972).
L. M. Kovba and V. I. Trunov, X-Ray Phase Analysis [in Russian], Izd. MGU, Moscow (1976).
A. I. Livshits, G. F. Lybzikov, and E. P. Zeer, in: Radiospectroscopy of the Solid State [in Russian], Vol. 1, Krasnoyarsk (1974), p. 177.
Yu. N. Moskvich, V. M. Buznik, P. P. Fedorov, and B. P. Sobolev, Kristallografiya,23, 416 (1978).
S. P. Gabuda, Yu. V. Gagarinskii, and S. A. Polishchuk, NMR in Inorganic Fluorides [in Russian], Atomizdat, Moscow (1978).
S. P. Gabuda, Yu. G. Kriger, and N. K. Moroz, Zh. Strukt. Khim.,17, 347 (1976).
E. P. Zeer, G. F. Lybzikov, V. V. Men'shikov, and Yu. A. Zagorodnyi, in: Radiospectroscopy of the Solid State [in Russian], Vol. 2, Krasnoyarsk (1976), p. 137.
B. Tanque, M. Vlasse, and J. Portier, Rev. Chim. Min.,10, 63 (1973).
Additional information
L. V. Kirenskii Institute of Physics, Siberian Branch, Academy of Sciences of the USSR. Translated from Zhurnal Strukturnoi Khimii, Vol. 24, No. 5, pp. 44–48, September–October, 1983.
Rights and permissions
About this article
Cite this article
Livshits, A.I., Buznik, V.M., Fedorov, P.P. et al. A19F NMR study of the anion mobility in lanthanum oxide fluorides. J Struct Chem 24, 689–693 (1983). https://doi.org/10.1007/BF00754801
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00754801