Abstract
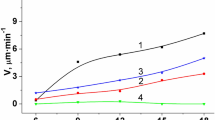
The revelation and morphology of dislocation etch pits as well as the rates of macroscopic dissolution and selective etching on the {1 0 0} plane of MgO crystals in aqueous solutions of various inorganic salts are investigated in relation to the nature and concentration of salt in solution and the etching temperature. It is found that addition of a salt generally facilitates etch pit formation and that the rates of surface dissolution and selective etching increase with additive concentration, etchant temperature and character and ageing of dislocations, while the etch pit morphology depends on the concentration and chemical nature of an impurity, etching temperature and the ageing of the dislocations. It is also observed that some fast etching solutions produce very shallow etch pits at screw dislocations. The results are discussed from a consideration of solution pH, standard electrode potentials of metals and stability of complexes present in solution. The importance of the surface entropy factor in revealing etch pits at screw dislocations is pointed out.
Similar content being viewed by others
References
K. Sangwal,J. Mater. Sci. 15 (1980) 237.
N. Cabrera andM. M. Levine,Phil. Mag. 1 (1956) 450.
N. Cabrera,J. Chim. Phys. 53 (1956) 675.
W. Schaarwächter,Phys. Stat. Solidi 12 (1965) 375.
Idem, ibid. 12 (1965) 865.
W. K. Burton, N. Cabrera andF. C. Frank,Phil. Trans. Roy. Soc. A243 (1951) 299.
W. G. Johnston, in “Progress in Ceramic Sciences” Vol. 2, edited by J. E. Burke (Pergamon Press, New York, 1962) p. 1.
R. B. Heimann, “Auflösung von Kristallen: Theorie und technische Anwendung” (Springer-Verlag, Wien, New York, 1975).
K. Sangwal andJ. N. Sutaria,J. Mater. Sci. 11 (1976) 2271.
K. Sangwal andT. C. Patel,Krist. Tech. 13 (1978) 1407.
K. Sangwal, T. C. Patel andM. D. Kotak,ibid. 14 (1979) 949.
Idem,J. Mater. Sci. 14 (1979) 1509.
Idem, ibid. 14 (1979) 1869.
K. Sangwal andS. K. Arora,ibid. 13 (1978) 1977.
E. M. Nadgornyi,Fiz. Tverd. Tela 5 (1963) 2723.
J. J. Gilman, W. G. Johnston andG. W. Sears,J. Appl. Phys. 29 (1958) 747.
A. R. Patel andJ. Koshy,Canad. Mineral 9 (1968) 539.
A. R. Patel andS. K. Arora,J. Phys. D: Appl. Phys. 7 (1974) 2301.
M. S. Joshi andM. A. Ittyachen,J. Appl. Phys. 40 (1969) 421;Ind. J. Pure Appl. Phys. 7 (1969) 624.
A. R. Patel andO. P. Bahl,Brit. J. Appl. Phys. 16 (1966) 169.
A. R. Patel andR. M. Chaudhri,Jap. J. Appl. Phys. 8 (1969) 677.
A. R. Patel andB. P. Agarwal,J. Phys. D: Appl. Phys. 4 (1971) 985.
B. Jeszenszky,Nature 181 (1958) 559.
K. Sangwal, M. Szurgot, J. Karniewicz andW. Kolasinski,J. Crystal Growth 58 (1982) 261.
K. Sangwal,J. Mater. Sci., in press.
K. Sangwal andA. A. Urusovskaya,Prag. Cryst. Growth Character in press.
N. N. Greenwood, E. J. F. Ross andB. P. Straughan, “Index of Vibration Spectra of Inorganic and Organometallic Compounds”, Vol. 1 (Butterworths, London, 1964).
C. K. Jörgensen, “Inorganic Complexes” (Academic Press, London, 1963).
H. D. Luedeman andE. U. Franck,Ber. Bunsenges. Phys. Chem. 71 (1967) 455.
S. N. Andreev andO. V. Sapozhnikova,Zh. Neorg. Khimii 10 (1965) 2538;13 (1968) 1548.
I. I. Antipova-Karataeva, Yu. A. Zolotov andI. V. Seryakova,ibid. 9 (1964) 1712.
K. Sangwal, N. L. Sizova andA. A. Urusovskaya,Krist. Tech. 12 (1977) 567.
I. Gajewska, S. Pietras, J. Rudzińska andA. Schellenberg (eds), “Poradnik Fizykochemiczny” (Physico-chemical Handbook) (Naukowotechniczne press, Warsaw, 1974).
J. Bjerrum, G. Schwarzenbach andL. G. Sinlén, “Stability Constants, Part 2: Inorganic Ligands” (Chemical Society, London, 1958).
P. Bennema, in “Industrial Crystallization 78”, edited by E. J. De Jong and S. J. Jančić (North-Holland, Amsterdam, 1979) p. 115.
H. J. Human, J. P. Van Der Eerden, L. A. M. J. Jetten andJ. G. M. Odekerken,J. Crystal Growth 51 (1981) 589.
P. Bennema andJ. P. Van Der Eerden,ibid. 42 (1977) 201.
P. Hartman, in “Crystal Growth: an Introduction”, edited by P. Hartman (North-Holland, Amsterdam, 1973) p. 363.
J. Gałecki, “Preparatyka Nieorganiczna” (Practical Inorganic Chemistry) (Naukowo-techniczne Press, Warsaw, 1964) p. 2423.
W. A. Weyl, in “Structure and Properties of Solid Surfaces” edited by R. Gomer and C. S. Smith (University of Chicago Press, Chicago, 1953) p. 147.
V. Hari Babu andK. G. Bansigir,J. Phys. Soc. Japan 23 (1967) 860.
Idem, J. Crystal Growth 2 (1968) 9.
K. Sangwal andA. A. Urusovskaya,ibid. 41 (1977) 216.
K. Sangwal, A. A. Urusovskaya andA. E. Smirnov,Ind. J. Pure Appl. Phys. 16 (1978) 501.
M. B. Ives,J. Phys. Chem. Solids 24 (1963) 275.
H. B. Huntington, J. E. Dickey andR. Thomson,Phys. Rev. 100 (1955) 1117.
J. D. Livingston, in “Direct Observation of Imperfections in Crystals” edited by J. B. Newkirk and J. H. Wernick (John Wiley, New York, London, 1962) p. 115.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sangwal, K. Dissolution kinetics of MgO crystals in aqueous acidic salt solutions. J Mater Sci 17, 3598–3610 (1982). https://doi.org/10.1007/BF00752203
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00752203