Abstract
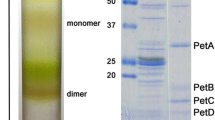
Exposure of antimycin-treated Complex III (ubiquinol-cytochromec reductase) purified from bovine heart mitochondria to [3H]succinic anhydride plus [35S]p-diazobenzenesulfonate (DABS) resulted in somewhat uniform relative labeling of the eight measured subunits of the complex by [3H]succinic anhydride. In contrast, relative labeling by [35S]DABS was similar to [3H]succinic anhydride for the subunits of high molecular mass, i.e., core proteins, cytochromes, and the iron-sulfur protein, but greatly reduced for the polypeptides of molecular mass below 15 kDa. With Complex III depleted in the iron-sulfur protein the relative labeling of core protein I by exposure of the complex to [3H]succinic anhydride was significantly enhanced, whereas labeling of the polypeptides represented by SDS-PAGE bands 7 and 8 was significantly inhibited. Dual labeling of the subunits of Complex III by14C- and3H-labeled succinic anhydride before and after dissociation of the complex by sodium dodecyl sulfate, respectively, was measured with the complex in its oxidized, reduced, and antimycin-inhibited states. Subunits observed to be most accessible or reactive to succinic anhydride were core protein II, the iron-sulfur protein, and polypeptides of SDS-PAGE bands 7, 8, and 9. Two additional polypeptides of molecular masses 23 and 12 kDa, not normally resolved by gel-electrophoresis, were detected. Reduction of the complex resulted in a significant change of14C/3H labeling ratio of core protein only, whereas treatment of the complex with antimycin resulted in decreases in14C/3H labeling ratios of core proteins I and II, cytochromec 1, and a polypeptide of molecular mass 13 kDa identified as an antimycin-binding protein.
Similar content being viewed by others
References
Anderson, S., DeBruijn, H. L., Coulson, A. R., Eperon, I. C., Sanger, F., and Young, I. G. (1982).J. Mol. Biol. 156, 683–717.
Ball, M. B., Bell, R. L., and Capaldi, R. A. (1977).FEBS Lett. 83, 99–102.
Baum, H., Silman, H. I., Rieske, J. S., and Lipton, S. H. (1967).J. Biol. Chem. 242, 4876–4887.
Bell, R. L., Sweetland, J., Ludwig, B., and Capaldi, R. A. (1979).Proc. Natl. Acad. Sci. USA 76, 741–745.
Clejan, L., Bosch, C. G., and Beattie, D. S. (1984).J. Biol. Chem. 259, 11169–11172.
Das Gupta, U., Wharton, D. C., and Rieske, J. S. (1979).J. Bioenerg. Biomembr. 11, 79–95.
DePierre, J. W., and Karnovsky, M. L. (1974).J. Biol. Chem. 249, 7111–7120.
D'Souza, M. P., and Wilson, D. F. (1982).J. Biol. Chem. 257, 11760–11766.
Engel, W. D., Michalski, C., and von Jagow, G. (1983).Eur. J. Biochem. 132, 395–402.
Furthmayr, H., and Timpl, R. (1971).Anal. Biochem. 41, 510–516.
Gellerfors, P., and Nelson, B. D. (1977).Eur. J. Biochem. 80, 275–282.
Ho, S. H. K. (1979). Ph.D. Dissertation, The Ohio State University, Columbus, Ohio.
Ho, S. H. K., and Rieske, J. S. (1983).Fed. Proc. 42 (No. 7), 1944 (Abstr. No. 1090).
Ho, S. H. K., Das Gupta, U., and Rieske, J. S. (1985).J. Bioenerg. Biomembr. 17, 263–276
Mendel-Hartvig, I., and Nelson, B. D. (1978).FEBS Lett. 92, 36–40.
Mendel-Hartvig, I., and Nelson, B. D. (1983).J. Bioenerg. Biomembr. 15, 27–36.
Nozaki, M., Mizushima, H., Horio, T., and Okunuki, K. (1958).J. Biochem. Tokyo 45, 815–823.
Reed, J., Reed, T. A., and Hess, B. (1978).Eur. J. Biochem. 91, 255–261.
Reed, J., Reed, T. A., and Hess, B. (1979).Proc. Natl. Acad. Sci. USA 76, 1045–1048.
Reynolds, J. A., and Tanford, C. (1970).J. Biol. Chem. 245, 5161–5165.
Rieske, J. S. (1967). InMethods in Enzymology (Colowick, S. P., and Kaplan, N. O., eds.), Vol. 10, Academic Press, New York, pp. 239–245.
Rieske, J. S. (1976).Biochim. Biophys. Acta 456, 195–247.
Rieske, J. S. (1980).Pharmacol. Ther. 11, 415–450.
Rieske, J. S., Baum, H., Stoner, C. D., and Lipton, S. H. (1967).J. Biol. Chem. 242, 4854–4866.
Rieske, J. S., Alben, J. O., and Liao, H. T. (1975). InElectron Transfer Chains and Oxidative Phosphorylation (Quagliariello, E., Papa, S., Palmieri, F., Slater, E. C., and Siliprandi, N., eds.), North-Holland/Elsevier, Amsterdam/New York, pp. 119–126.
Rodbard, D., and Chrambach, A. (1971).Anal. Biochem. 40, 95–134.
Schägger, H., von Jagow, G., Borchart, U., and Machbidt, W. (1983).Hoppe-Seyler's Z. Physiol. Chem. 364, 307–311.
Sidhu, A., and Beattie, D. S. (1982).J. Biol. Chem. 257, 7879–7886.
Smith, R. J., and Capaldi, R. A. (1977).Biochemistry 16, 2629–2633.
Smith, R. J., Capaldi, R. A., Muchmore, D., and Dahlquist, F. (1978).Biochemistry 17, 3719–3723.
Trumpower, B. L., and Katki, A. (1975).Biochemistry 14, 3635–3642.
Trumpower, B. L., and Edwards, C. A. (1979).J. Biol. Chem. 254, 8697–8706.
Trumpower, B. L., Edwards, C. A., Katki, A., and Ohnishi, T. (1979). InMembrane Bioenergetics (Lee, C. P., Shatz, G., and Ernster, L., eds.), Addison-Wesley, London, pp. 217–228.
Wakabayashi, T., Senior, A. E., Hatase, O., Hayashi, H., and Green, D. E. (1972).J. Bioenerg. 3, 339–344.
Wakabayashi, S., Takeda, H., Matsubara, H., Kim, C., and King, T. E. (1982).J. Biochem. 91, 2077–2085.
Weber, K., and Osborn, M. (1969).J. Biol. Chem. 244, 4406–4412.
Yamanaka, T., Mizushima, H., Nozaki, M., Horio, T., and Okunuki, K. (1959).J. Biochem. Tokyo 46, 121–132.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ho, S.H.K., Rieske, J.S. Differential labeling of the subunits of respiratory Complex III with [3H]succinic anhydride, [14C]succinic anhydride, andp-diazobenzene-[35S]sulfonate. J Bioenerg Biomembr 17, 333–348 (1985). https://doi.org/10.1007/BF00743107
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00743107