Abstract
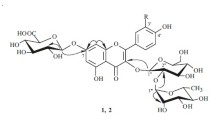
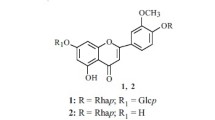
Two new glycosides of limocitrin have been isolated from the epigeal part ofHaplophyllum perforatum (M. B.) Kar. et Kir. On the basis of chemical transformations and spectral characteristics the structures of the substances isolated have been established as 7-(6″-acetyl-β-D-glucopyranosyloxy)-3,4′,5-trihydroxy-3′,8-dimethoxyflavone and 7-[0-α-L-rhamnopyranosyl-(1 → 2)-β-D-glucopyranosyloxy]-3,4′,5-trihydroxy-3′,8-dimethoxyflavone.
Similar content being viewed by others
Literature cited
É. Kh. Batirov and V. M. Malikov, Khim. Prir. Soedin., 330 (1980).
É. Kh. Batirov, V. M. Malikov, and M. E. Perel'son, Khim. Prir. Soedin., 304 (1981).
T. J. Mabry, K. R. Markham, and M. V. Thomas, The Systematic Identification of Flavonoids, Springer, New York (1970), p. 41.
T. J. Mabry and K. R. Markham, “Mass spectrometry of flavonoids,” in: The Flavonoids, J. B. Harborne, T. J. Mabry, and H. Mabry, eds., Chapman and Hall, London (1975), p. 90.
M. Goudard, J. Farve-Bovnin, P. Lebreton, J. Chopin, Phytochemistry,17, 145 (1978).
R. M. Horovitz and B. Gentili, J. Org. Chem.,26, 2899 (1961).
H. Jay, A. Hasan, B. Voirin, and M.-R. Viricel, Phytochemistry,17, 827 (1978).
J. M. van der Veen, J. Org. Chem.,28, 564 (1963).
R. Higuchi, and D. M. X. Donnelly, Phytochemistry,17, 787 (1978).
S. Hakomori, J. Biochem. (Tokyo),55, 205 (1964).
K. Miyahara and T. Kawasaki, Chem. Pharm. Bull.,22, 1407 (1974).
G. G. Zapesochnaya, Khim. Prir. Soedin., 695 (1982).
J. B. Harborne, Phytochemistry,20, 1117 (1981).
Additional information
Institute of the Chemistry of Plant Substances, Academy of Sciences of the Uzbek SSR, Tashkent. Translated from Khimiya Prirodnykh Soedinenii, No. 2, pp. 192–196, March–April, 1985.
Rights and permissions
About this article
Cite this article
Yuldashev, M.P., Batirov, É.K. & Malikov, V.M. Flavonoids ofHaplophyllum perforatum. New glycosides of limocitrin. Chem Nat Compd 21, 179–182 (1985). https://doi.org/10.1007/BF00714907
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00714907