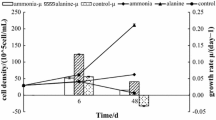
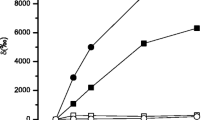
Summary
The proportion of N derived from N2 fixation for 99 strains ofAzolla spp. (comprising all known species) in the presence of ammonium (40 mg/1) was assessed using a15N-dilution technique. The percentage of N derived from air varied from 29.5% to 79.9%. Although the N concentration ofAzolla spp. was not correlated with fertilizer N, it correlated fairly well with N2 fixation. Regression analysis suggests that the N yield ofAzolla spp. is more dependent on N2 fixation than on ammonium assimilation. The high correlation between N yield and isotopically determined, fixed N2 indicates that the N yield could be used as a parameter in the selection ofAzolla spp. strains that are capable of maintaining high N2 fixation in the presence of a high level of ammonium.
Similar content being viewed by others
References
Eady R (1981) Regulation of nitrogenase activity. In: Gibson AH, Newton WE (eds) Current perspectives in nitrogen fixation. Proc 4th int symp on nitrogen fixation, Canberra, 1–5 Dec 1980. Australian Academy of Science, Canberra, pp 172–181
Fiedler R, Proskch G (1975) The determination of nitrogen-15 by emission and mass spectrometry in biochemical analysis: A review. Anal Chim Acta 78:1–62
Hardarson G, Zapata F, Danson SKA (1984) Field evaluation of symbiotic nitrogen fixation by rhizobial strains using15N methodology. Plant and Soil 82:369–375
Hartmann A, Fu HA, Song SD, Burris RH (1985) Comparison of nitrogenase regulation inA. brasilense, A. lipoferum andA. amazonense. In: Klingmüller W (ed)Azospirillum 3: Genetic, physiology, ecology. Proc Bayreuth Azospirillum Workshop 3rd, Springer, Berlin Heidelberg New York, pp 116–126
Hopmans P, Douglas LA, Chalk PM (1984) Effects of soil salinity and mineral nitrogen on acetylene reduction activity inTrifolium subterraneum L. Aust J Agric Res 35:9–15
Kallio S (1978) On the effects of forest fertilizers on nitrogenase activity in two subarctic lichens. In: Granhall U (ed) Environmental role of nitrogen fixing blue-green algae and asymbiotic bacteria. Liber Tryck, Stockholm, Ecol Bull (Stockh) 26:217–224
Klugkist J, Haaker H (1984) Inhibition of nitrogenase activity by ammonium chloride inAzotobacter vinelandii. J Bacteriol 157:148–151
Kumarasinghe KS, Zapata F, Eskew DL, Arrilaga JL, Danso SKA (1985)Azolla Anabaena associations:15N substratum labelling technique to assess N2-fixation and its potential as biofertilizer for rice. In: Ssali H, Keya SO (eds) Proc first conference of the African Association for Biological Nitrogen Fixation (AABNF) held in Nairobi, Kenya, 23–27 July 1984 MIRCEN, Nairobi, pp 454–464
Lumpkin TA (1987) Collection, maintenance and cultivation ofAzolla. In: Elkan GH (ed) Symbiotic nitrogen fixation technology. Dekker, New York Basel, pp 55–94
Mackerras AH, Smith GD (1986) Evidence for direct repression of nitrogenase by ammonia in the cyanobacteriumAnabaena cylindrica. Biochem Biophys Res Commun 134:834–844
Ohmori M, Hattori A (1978) Transient change in the ATP pool ofAnabaena cylindrica associated with ammonium assimilation. Arch Microbiol 117:17–20
Peters GA, Ito O (1984) Determining nitrogen fixation and elemental nitrogen input inAzolla grown with or without combined nitrogen sources: Keeping the acetylene reduction assay in the proper perspective. Dev Plant Soil Sci 13:29–44
Peters GA, Toia Jr RE, Evans WR, Crist DK, Mayne BC, Pocle RE (1980) Characterization and comparisons of five N2-fixingAzolla Anabaena associations: I. Optimization of growth conditions for biomass increase and N content in a controlled environment. Plant Cell Environ 3:261–269
Peters GA, Ito O, Tyagi VVS, Mayne BC, Kaplan D, Calvert HE (1981) Photosynthesis and N2-fixation in theAzolla-Anabaena symbiosis. In: Gibson AH, Newton WE (eds) Current perspectives in nitrogen fixation. Proc 4th int symp on nitrogen fixation, Canberra, 1–5 Dec 1980, Australian Academy of Science, Canberra, pp 121–124
Rai AN, Rowell P, Stewart WDP (1984) Evidence for an ammonium transport system in free-living and symbiotic cyanobacteria. Arch Microbiol 137:241–246
Statistical Analysis System (1982) SAS user's guide, Statistics. SAS Institute Inc, North Carolina, pp 39–90
Van Hove C, de Waha Baillonville T, Godard P, Mai Kodomi Y, Sanginga N (1987)Azolla collection and selection. In:Azolla utilization. Proc Workshop onAzolla use, Fuzhou, China, 31 March–5 April 1985. Int Rice Res Inst (Los Baños), pp 77–88
Yoneyama T, Ladha JK, Watanabe I (1987) Nodule bacteroids andAnabaena: Natural15N enrichment in the legume-rhizobium andAzolla Anabaena symbiosis systems. J Plant Physiol 127:251–259
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Okoronkwo, N., van Hovel, C. & Eskew, D.L. Evaluation of nitrogen fixation by different strains of theAzolla-Anabaena symbiosis in the presence of a high level of ammonium. Biol Fert Soils 7, 275–278 (1989). https://doi.org/10.1007/BF00709661
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00709661