Abstract
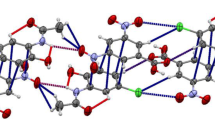
The 1,4-dichloro-(1) and 1,4-dibromo-(2) derivatives of naphthalene-2,3-diol crystallise in structures containing acetic acid or dioxane solvent molecules. X-ray crystallographic examination of the compound formed between1 and dioxane is reported here [(C10H6Cl2O2)2·(C4H8O2),P21/c,a=12.358(3),b=4.9930(7),c=19.167(4) Å,β=96.09(1)0,Z=2,R=0.035] and this structure is analysed in crystal engineering terms. The compound is a co-crystalline material involving two types of hydrogen bonding: one phenolic group participates in a
chain, while the second phenolic group hydrogen bonds to a dioxane. Extension of the hydrogen bonding network through the second dioxane oxygen results in heavily corrugated layers. Neighbouring layers interact by a combination of aromatic face-face and edge-face interactions similar to a partial coronene-type γ packing to complete the structure.
Similar content being viewed by others
References
S.C. Wallwork and H.M. Powell:J. Chem. Soc. Perkin Trans. 2 641 (1980).
R. Perrin, R. Lamartine, M. Perrin and A. Thozet, in:Organic Solid State Chemistry, Ed. G.R. Desiraju, Ch. 8, pp. 271–329, ‘Studies in Organic Chemistry: 32’, Elsevier, Amsterdam (1987).
D.D. MacNicol in:Inclusion Compounds, Vol. 1, Eds. J.L. Atwood, J.E.D. Davies and D.D. MacNicol, Ch. 1, pp. 1–45, Academic Press, London (1984).
A.T. Ung, R. Bishop, D.C. Craig, I.G. Dance and M.L. Scudder:J. Chem. Soc., Chem. Commun. 322 (1993).
R. Bishop, D.C. Craig, I.G. Dance, M.L. Scudder and A.T. Ung:Mol. Cryst. Liq. Cryst. 240, 113 (1994).
A.T. Ung, R. Bishop, D.C. Craig, I.G. Dance and M.L. Scudder:Chem. Mater. 6, 1269 (1994).
A.T. Ung, D. Gizachew, R. Bishop, D.C. Craig, I.G. Dance and M.L. Scudder:J. Am. Chem. Soc. (accepted for publication).
G.R. Desiraju:Crystal Engineering: The Design of Organic Solids. Elsevier, Amsterdam (1989). (a) Ch. 4, pp. 94–96.
R. Willstätter and J. Parnas:Chem. Ber. 40, 3971 (1907). [See footnote on p. 3974.]
T. Zincke and K. Fries:Annalen 334, 342 (1904).
R.M. Herath Banda, I.G. Dance, T.D. Bailey, D.C. Craig and M.L. Scudder:Inorg. Chem. 28, 1862 (1989).
J. de Meulenaer and H. Tompa:Acta Crystallogr. 19, 1014 (1965).
P. Main, S.J. Fiske, S.E. Hull, L. Lessinger, G. Germain, J.-P. Declercq and M.M. Woolfson: MULTAN80, Universities of York, England and Louvain, Belgium (1980).
W.R. Busing, K.O. Martin and H.A. Levy: ORFLS, Oak Ridge National Laboratory, Tennessee, U.S.A. (1962). BLOCKLS is a local version of this program.
A.D. Rae: RAELS, A Comprehensive Constrained Least-Squares Refinement Program, University of New South Wales (1989).
A. Gavezzotti and G.R. Desiraju:Acta Cystallogr., Sect. B 44, 427 (1988).
K.C. Pich, R. Bishop, D.C. Craig and M.L. Scudder:J. Incl. Phenom. 18, 149 (1994).
For example: I. Csöregh, E. Weber, L.R. Nassimbeni, O. Gallardo, N. Dörpinghaus, A. Ertan and S.A. Bourne:J. Chem. Soc., Perkin Trans. 2 1775 (1993).
J.C. Barnes:Acta Crystallogr., Sect. C 44, 118 (1988).
J.C. Barnes, J.D. Paton and C.S. Blyth:Acta Crystallogr., Sect. C 46, 1183 (1990).
T. Gramstad, S. Husebye and K. Maartmann-Moe:Acta Chem. Scand., Ser. B 39, 767 (1985).
Author information
Authors and Affiliations
Additional information
Supplementary Data relating to this article are deposited with the British Library as supplementary publication No. SUP 82184 (11 pages).
Rights and permissions
About this article
Cite this article
Ahn, P.D., Bishop, R., Craig, D.C. et al. Structure of the co-crystalline solid formed between 1,4-dichloronaphthalene-2,3-diol and dioxane. J Incl Phenom Macrocycl Chem 20, 267–276 (1994). https://doi.org/10.1007/BF00708772
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00708772